- You must be logged in to add items to the cart.
$495.00
Description
This prescription GLP-1 receptor agonist comes as a prefilled pen for subcutaneous use in adult weight management programs. Clinics use this therapy within structured protocols that include nutrition and activity counseling. It is available through US distribution to simplify ordering and coordination for licensed practices.
What Saxenda Is and How It Works
Med Wholesale Supplies serves licensed clinics and healthcare professionals with authentic, brand-name medical products sourced through vetted distributors.
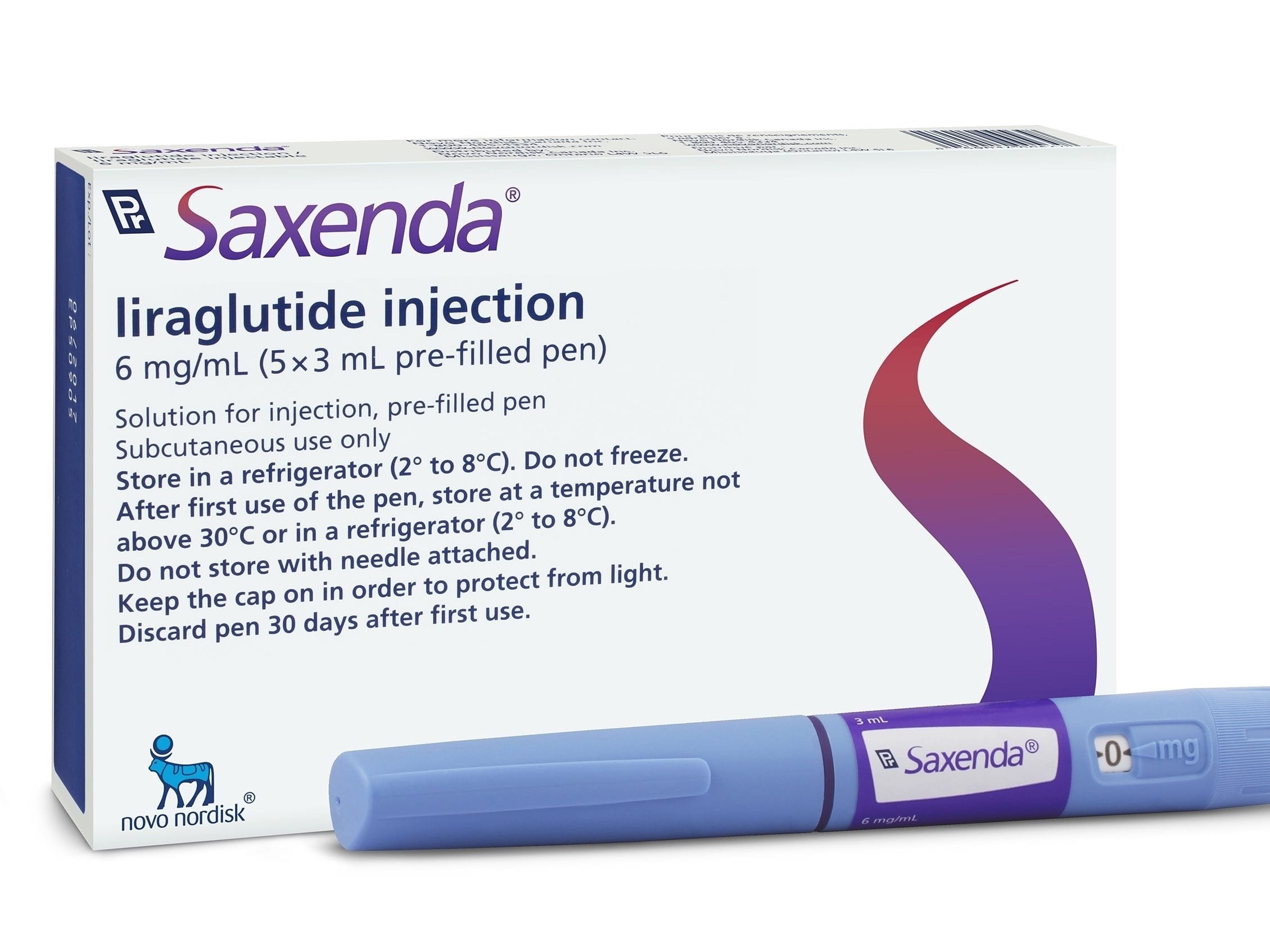
Saxenda® contains liraglutide, a GLP-1 analogue supplied in a ready-to-use, multi-dose pen for subcutaneous administration. This class supports appetite regulation and aligns with professionally supervised weight management pathways. The pen format helps standardize daily administration in clinical settings and reduces preparation time compared with vial-and-syringe workflows.
The mechanism of action mirrors endogenous GLP-1 activity, which can promote satiety cues and support adherence to structured nutrition plans. Providers typically integrate this preparation alongside monitoring, counseling, and follow-up visits. As with all prescription injectables, administration should follow the approved labeling and institutional protocols. In multidisciplinary programs, clinicians often position this therapy as liraglutide used for chronic weight-management care combined with behavioral interventions. For additional context on program design, see our Clinical Overview.
Professional Applications
Clinics deploy this GLP-1 pen within medically supervised weight-management services. Typical workflows include initial evaluation, risk assessment, informed consent, patient education, and scheduled reviews. Teams use it in conjunction with nutrition counseling and activity plans, reinforcing consistent habits over time.
Administration occurs subcutaneously using pen-injector technique. Clinicians use standard Saxenda injection sites such as the abdomen, thigh, or upper arm, following rotation practices consistent with routine subcutaneous care. This format suits in-office demonstration and supports at-home continuity when prescribed and supervised by qualified providers. To support staff training and patient education frameworks, see Weight Loss Injections.
Programs may embed this therapy within broader service lines, including nutrition classes, metabolic coaching, and periodic reassessment visits. Documentation often includes baseline metrics, tolerability checks, and ongoing adherence support. Clinics also review contraindications and past medical history before initiating therapy.
Key Features
- Ready-to-use pen: multi-dose, subcutaneous administration.
- GLP-1 analogue class: supports structured weight-management protocols.
- Dial-a-dose design: facilitates incremental titration per label.
- Prefilled solution: reduces compounding steps and waste.
- Clinic-friendly handling: compact footprint for medication storage.
- Sterile, manufacturer-sealed primary packaging.
- Clear labeling: lot and expiration visible for inventory control.
- Single-patient use: aligns with infection control standards.
- Education support: suitable for in-office technique demonstration.
Benefits in Practice
This preparation integrates smoothly into existing visit templates and staff competencies. The pen device helps standardize administration steps and supports consistent documentation. Clinics can incorporate it into multidisciplinary pathways, ensuring clear division of tasks among prescribers, nurses, and educators.
Operationally, the pen design may reduce training time compared with vial workflows, and it can streamline room turnover. Inventory teams appreciate the labeled, sealed format with visible identifiers for lot and expiry checks. These attributes help clinics maintain predictable processes while focusing on counseling and engagement.
For context on GLP-1 therapy selection and program design, see our GLP-1 Overview. Clinics should align use with local regulations, payer requirements where applicable, and internal clinical governance.
Composition & Ingredients
Active ingredient: liraglutide. The solution is formulated for subcutaneous administration in a prefilled pen. In professional practice, the common maintenance target per label is the Saxenda 3 mg dose. Initiation and adjustments follow the approved prescribing information and clinic protocols. Excipients follow the manufacturer’s labeled formulation and typically include buffering agents, preservative, and water for injection.
Always review the current label and your institutional policies before use. For safety topics commonly discussed in clinics, see Safety Considerations.
Packaging & Supply
This line is supplied as prefilled pen-injectors designed for multi-dose use by a single patient under a licensed prescriber’s supervision. The labeled concentration is Saxenda 6 mg/ml. Each pen supports daily use with an adjustable dial, enabling stepwise titration per the official label. Primary packaging is manufacturer-sealed for integrity.
Cartons and pens display product identifiers, lot numbers, and expiration dates for inventory control. Packaging components may include instructions for use and safety information. For clinics evaluating other configurations, see the International Pack available from our catalog.
Ordering & Logistics
Access is limited to licensed healthcare entities. Please create or verify your account to request allocation and to view contract options. Orders can be placed through our secure portal after credential review and approval. Sign in to see availability aligned to your verified practice profile.
Our team coordinates temperature-controlled handling when required and tracked US delivery. We provide shipment confirmations with lot details visible upon receipt so your team can complete intake and storage checks efficiently.
For broader therapeutic selections, browse Pharmaceuticals across categories relevant to your practice type.
Comparable Products
Clinics that staff GLP-1 programs often evaluate molecules, dosing schedules, and device formats to match patient needs and follow-up capacity. If your workflow calls for alternatives within the weight-management space, explore our Weight Loss category for options that align with your protocols and visit cadence.
Practices may also review alternative packaging or regional versions. To survey the broader line within our catalog, visit the brand Brand Page for currently listed configurations.
Pricing & Access
Sign in to view live pricing. Volume and contract tiers are available for qualified accounts, with allocations aligned to verified use cases. Your account manager can assist with quotes for recurring schedules, consolidated shipments, and multi-site purchasing if applicable.
Availability & Substitutions
Inventory fluctuates based on manufacturer release cycles and regional demand. If a configuration is temporarily unavailable, your account manager can discuss clinically appropriate substitutions within your protocols. Confirm any therapeutic changes with your prescribers before implementation and document transitions according to your policy.
When considering alternatives, review label differences, device training needs, and appointment cadence. Align operational choices with staffing, patient follow-up capacity, and program outcomes you track internally.
Authoritative Sources
Sign in to verify your clinic account and request allocation for your next procurement window.
Frequently Asked Questions
Licensing and Prescribing Requirements
This medication is a prescription GLP-1 therapy intended for use under a licensed prescriber’s supervision. It should be administered by qualified healthcare personnel following the official label and your organization’s policies. Ensure your clinic’s account is verified before ordering. This product is not intended for consumer self-administration or retail sale and must be used within a structured medical program that includes appropriate evaluation and follow-up.
Training and Administration Workflow
Most clinics incorporate device demonstration, technique coaching, and follow-up assessments into their workflow. A brief in-office demonstration helps confirm pen setup, site rotation guidance, and safe disposal practices. Educational materials can be provided during onboarding, with reinforcement at subsequent visits. Incorporate monitoring for tolerability and reinforce lifestyle counseling as part of your clinic’s protocol and documentation standards.
Storage and Handling Guidance
Follow the manufacturer’s label for storage and stability instructions, and maintain chain-of-custody documentation per your policy. Use appropriate protection during transit and intake, then place into your medication storage according to local regulations. Do not use any unit with compromised packaging or obscured lot and expiry details. If questions arise about excursions or integrity, quarantine the unit and consult the label or manufacturer support channels.
Comparing Options in GLP-1 Care
Clinics often evaluate daily pen-injectors against weekly alternatives during therapy selection. Discussions of Saxenda v Wegovy typically focus on molecule, schedule, device familiarity, and practice capacity for follow-up. Choose according to your protocols, staffing, and patient adherence considerations. Provide clear education, monitor tolerability, and review program outcomes when determining which GLP-1 path fits your service line best.
Generic and Therapeutic Equivalence
There is no approved Saxenda generic currently supplied in our catalog. While liraglutide is the active ingredient, branded device, formulation, and labeling distinguish this product. If you require alternatives, consult your prescribers about therapeutic substitutions within your program, and ensure any transition includes education, monitoring, and documentation aligned to your clinic’s policy and the relevant label.
Access to Official Labeling
Clinicians should review Saxenda prescribing information for dosing, administration, contraindications, and warnings. Use the current U.S. label as the primary source, and align clinic protocols with those directions and local regulations. Keep a copy accessible in your medication area and within electronic references. For complex cases or questions, consult the manufacturer’s medical information service or pharmacy support for clarification.
Dose Planning and Follow-Up
Establish a structured titration and review schedule per the official label and your clinical governance. Many programs include early follow-up to assess tolerability, reinforce nutrition guidance, and adjust the plan as needed. Incorporate adverse event monitoring and document site rotation practices. Coordinate with your pharmacy or nursing leadership to ensure consistent training, clear patient instructions, and reliable supply visibility across appointments.
Specifications
- Main Ingredient: Liraglutide
- Manufacturer: NOVO NORDISK
- Drug Class:
- Generic Name: Liraglutide
- Package Contents: 3 mL x 5 Pre-Filled Syringes
- Storage Requirements: Room Temperature (2℃~25℃)
- Main Usage: Weight Loss
About the Brand
SAXENDA®
Here to help
Questions about ordering, delivery or products? You can email our team here or call now at 1-800-630-9757 and be connected with your dedicated Account Manager
Related Products
NEAUVIA™ ORGANIC INTENSE LIPS
RejuvaNAD+
Related Articles
Osteoporosis Bone Building Drugs: Anabolic Options for Clinics
Key Takeaways Define the goal: distinguish bone-forming therapies from antiresorptives. Sequence thoughtfully: transitions can matter…
Md Ceuticals Sunscreen Clinical Guide for Clinic Selection
Key Takeaways Md ceuticals sunscreen can be assessed like any clinical sunscreen: verify claims on…
Sculptra Side Effects: What Clinics Should Screen and Track
Key Takeaways Tracking Sculptra side effects starts with clear expectations, tight documentation, and consistent follow-up.…
Hyaluronidase for Lip Filler: Clinical Workflow Essentials
Key Takeaways Hyaluronidase for lip filler is typically used to reverse hyaluronic acid gel in…
What Is Mesotherapy? Clinical Uses, Risks, and Workflow
Key Takeaways Define the technique: what is mesotherapy refers to superficial multi-injection delivery of small…
Juvederm Before And After Photos Documentation Guide For Clinics
Key Takeaways Standardize images: Control pose, lighting, lens, and distance. Document context: Record product, lot,…
Esthetician License Requirements for Med Spa Clinic Teams
Key Takeaways Verify licensure status: confirm active status with the state board. Match role to…
Esthetician vs Aesthetician: Clinic Role and Credential Guide
Key Takeaways Spelling varies by region and employer style guides. Licensing rules come from state…