Saxenda for weight loss can fit a clinic program when you treat it as both a clinical and operational decision. It is a liraglutide-based GLP-1 receptor agonist (an appetite-regulating hormone mimic) given by daily injection, so success depends on more than eligibility alone. For teams reviewing clinic-facing considerations, the real work is patient selection, contraindication screening, pen teaching, tolerability follow-up, and clean documentation from baseline onward.
Key Takeaways
- Use label-based eligibility and a structured baseline screen before initiation.
- Expect gastrointestinal effects to drive many early questions and discontinuations.
- Standard pen training and missed-dose protocols reduce avoidable confusion.
- Monitor for serious warning signs, not just routine nuisance symptoms.
- Compare daily workflow burden with other GLP-1 options before standardizing a protocol.
This resource is written for licensed clinics and healthcare professionals.
How Saxenda Fits in Weight-Management Care
Saxenda for weight loss is the brand formulation of liraglutide used in chronic weight-management care under current labeling. At a high level, it works by activating GLP-1 pathways that can reduce appetite, increase satiety, and slow gastric emptying. That can support lower calorie intake, but it does not replace nutrition, activity, or longitudinal follow-up.
Why this matters operationally is simple. A daily injectable therapy creates more training needs and more early friction than many oral agents. Patients may qualify on paper yet still struggle with self-administration, schedule consistency, or tolerability. Clinics that ignore those points usually see more messages, more missed follow-ups, and less consistent documentation.
It also helps to frame the therapy correctly from the first visit. This is not a cosmetic quick fix or a short burst intervention. It belongs in a structured weight-management plan with clear clinical goals, counseling expectations, and a process for reviewing benefit versus burden over time.
For broader category context, the site’s Weight Loss hub can help teams map related topics without turning the encounter into a product discussion.
Who May Be a Fit for Therapy
Eligibility is usually label-based, not preference-based. In practice, clinics start by confirming whether the patient meets current criteria for chronic weight management, then screen for contraindications, prior treatment history, and readiness for a daily injection routine.
For adult use, labeling has generally centered on obesity, or overweight with at least one weight-related comorbidity. Exact criteria, age limits, and payer rules can change, so staff should verify current labeling and coverage requirements rather than rely on memory or an old dosing sheet. If the clinic sees adolescents or complex endocrine patients, those pathways usually need extra review and protocol clarity.
Good patient selection is also practical. Someone may meet the medical threshold yet still struggle with self-injection, follow-up reliability, severe baseline nausea, unstable meal patterns, or competing medications that complicate monitoring. Those issues do not automatically rule treatment in or out, but they do affect whether the plan is realistic and safe.
Baseline assessment points
- Weight history: prior attempts, rate of gain, and current treatment goals.
- Related conditions: cardiometabolic risk, sleep apnea, fatty liver, or other weight-linked disease.
- Contraindication screen: boxed warning review, endocrine history, and pregnancy status when relevant.
- Medication review: other GLP-1 agents, diabetes therapies, and drugs affected by slowed gastric emptying.
- Behavioral fit: injection readiness, health literacy, and follow-up reliability.
- Baseline metrics: weight and other clinic-defined starting measures for later comparison.
Quick tip: Use one baseline template so every prescriber and nurse captures the same exclusion and monitoring points.
Expectation setting belongs here, not after the first complaint call. Patients need to understand that response is variable, early adverse effects are common, and follow-up is part of treatment rather than an optional extra. Clinics should also decide in advance how they will document nonresponse, poor adherence, or inability to tolerate escalation according to the official prescribing information.
Safety Signals, Side Effects, and When to Reassess
The most common adverse effects are gastrointestinal. Nausea, vomiting, diarrhea, constipation, abdominal discomfort, and reduced appetite are often the issues patients notice first, and these are a major reason clinics need early check-ins. In day-to-day practice, tolerability may matter more than theory.
When staff ask about the ‘biggest side effect,’ the practical answer is usually nausea or broader GI intolerance rather than one dramatic event. Mild to moderate symptoms are common enough to affect hydration, nutrition, confidence, and adherence. That is why pre-start counseling should explain what is common, what is temporary, and what needs a same-day call.
Serious risks sit in a different category. Clinics should review current labeling for pancreatitis warnings, gallbladder disease, dehydration-related renal complications, hypersensitivity, mood changes or suicidal thinking, and the boxed warning tied to thyroid C-cell tumor findings in rodents. The point is not to overstate danger. The point is to separate routine monitoring from escalation triggers.
Red flags that should trigger prompt clinical review
- Persistent severe abdominal pain: especially if it is new, intense, or worsening.
- Repeated vomiting or poor intake: dehydration can quickly complicate care.
- Gallbladder-type symptoms: right upper abdominal pain, fever, or jaundice need assessment.
- Allergic features: rash, swelling, or breathing difficulty need urgent review.
- Marked glycemic change: this matters most with other glucose-lowering therapies.
- Mood or behavior change: new concerning symptoms should not be dismissed.
Interactions deserve a practical review as well. Because liraglutide slows gastric emptying, some oral medications may need closer attention around initiation or dose escalation. Patients using insulin or insulin secretagogues may also require more careful oversight because glucose patterns can shift. A full medication reconciliation before initiation and again at follow-up is more useful than a one-time checkbox.
The main ‘do nots’ are straightforward. Do not combine therapies casually across the same class or with other liraglutide-containing products. Do not minimize persistent GI symptoms that threaten hydration or nutrition. Do not continue with vague documentation when a patient is clearly struggling with administration, tolerance, or follow-up engagement.
Internet rumors also need context. Shortages, backorders, or manufacturer portfolio changes are not the same as a blanket regulatory withdrawal. If a patient or staff member asks whether the drug has been taken off the market, verify the answer through current FDA information and official manufacturer communications rather than forum posts.
Injectable procurement should rely on vetted distributors and verified supply channels.
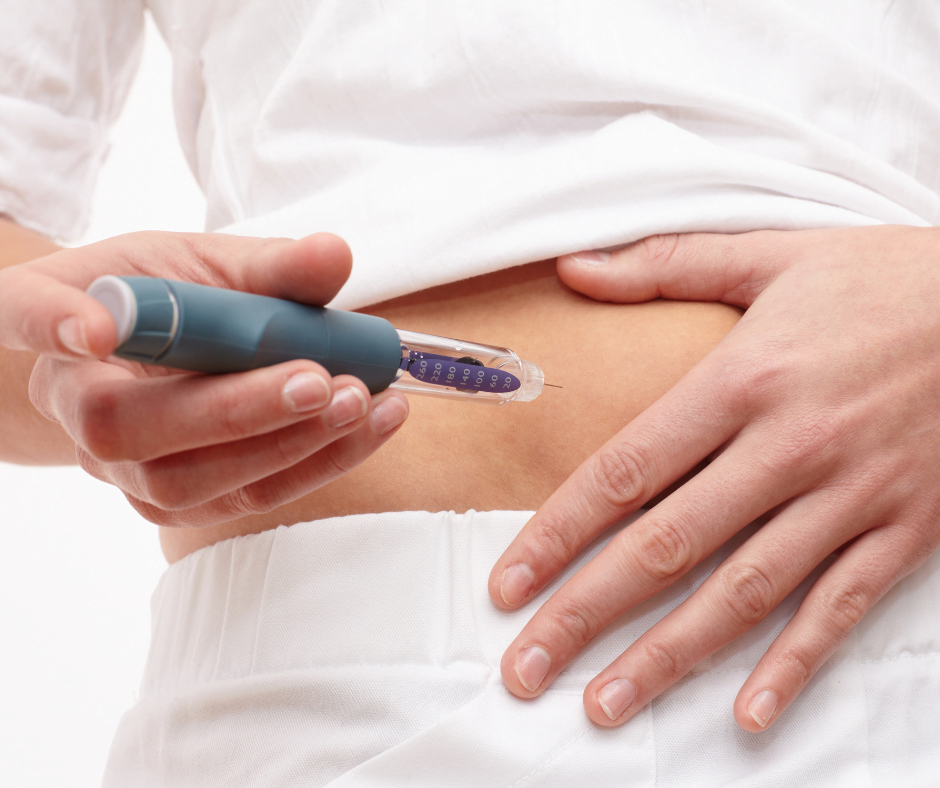
Administration Counseling and Early Follow-Up
Saxenda for weight loss requires more than a prescription handoff. Staff should confirm that the patient understands daily administration, titration according to current labeling, injection-site rotation, missed-dose instructions, and when to call the clinic about intolerance. Labeling uses a gradual escalation schedule rather than starting at maintenance exposure, largely to improve gastrointestinal tolerance.
Pen teaching should be simple and repeatable. A brief nurse-led or pharmacist-led demonstration, followed by patient return demonstration, can uncover confusion about needle attachment, priming steps, dose dialing, site selection, and disposal before the first problem call. That process is often more valuable than giving a long handout with no teach-back.
The first follow-up window is mainly about tolerance, adherence, and expectations. Many patients focus only on the scale. Clinics do better when they also ask about nausea burden, hydration, bowel changes, meal patterns, mood, and injection confidence. Those factors often explain whether a treatment plan is workable.
Why it matters: Daily injection burden and early GI symptoms can derail treatment even when eligibility is clear.
Missed-dose questions should be handled with the official prescribing information in front of you. Re-start rules and interruption thresholds should not be improvised from memory. The same applies to storage and handling. Follow current label instructions for unopened product, in-use product, temperature limits, and beyond-use timing rather than borrowing habits from another injectable.
For teams mapping category options, the site’s Weight Loss Products collection can help frame broader inventory discussions.
Comparing Daily GLP-1 Workflow Demands
Saxenda for weight loss is not just another name in the same class. Its daily injection schedule changes staffing, adherence, and follow-up demands compared with weekly GLP-1 options. That difference can shape patient preference, training time, refill management, and the number of early troubleshooting calls a clinic should expect.
From an operational standpoint, comparison should start with workflow rather than marketing claims. Ask which option matches the patient’s ability to self-manage injections, which one aligns with documentation and payer requirements, and how each choice affects early follow-up burden. If a clinic is standardizing forms or protocols, those details matter more than brand familiarity.
It also helps to separate class overlap from product-specific detail. Liraglutide-based therapy may share broad GLP-1 counseling themes with other agents, but device steps, titration rules, and stock handling still need product-specific verification every time. Head-to-head assumptions should be made cautiously unless they are supported by appropriate evidence and current labeling.
If your team needs broader operational reading, the Clinic Operations hub is a useful place to align protocols across prescribers and support staff.
Clinic Workflow Snapshot
A simple workflow prevents most avoidable gaps. Saxenda for weight loss usually works best in clinics that treat initiation as a repeatable process rather than a one-off prescription.
- Verify fit: confirm label-based indication, exclusions, and concurrent therapy review.
- Document baseline: record weight history, related disease, counseling, and starting symptoms.
- Confirm sourcing: retain lot and supplier data per clinic policy and current regulations.
- Teach the pen: standardize demonstration, return demo, and sharps disposal instructions.
- Check storage: follow official handling rules for refrigerated and in-use pens.
- Schedule follow-up: review tolerance, hydration, adherence, and review triggers early.
- Record outcomes: note response, adverse effects, and next-step decisions consistently.
Clinics should also define what counts as a review point versus a stop point before the first prescription goes out. Examples may include persistent intolerance, inability to follow administration steps safely, new contraindication findings, or repeated disengagement from follow-up. A preset escalation path reduces mixed messages across different staff members.
Brand-name procurement should be tied to documented sourcing records.
Authoritative Sources
- Current FDA prescribing information for Saxenda
- NCBI Bookshelf clinical review of liraglutide
- Official manufacturer pen-use instructions
Used well, this therapy can be part of a structured weight-management workflow. The clinic value comes from disciplined selection, realistic counseling, careful monitoring, and dependable documentation rather than from the drug name alone.
This content is for informational purposes only and is not a substitute for professional medical advice.