Key Takeaways
- Check the label: Storage limits vary by product and presentation.
- Control excursions: Track time out of refrigeration and peak temperature.
- Separate dates: Expiration, in-use limits, and BUD are not identical.
- Quarantine when unsure: Hold product and verify with authoritative sources.
Overview
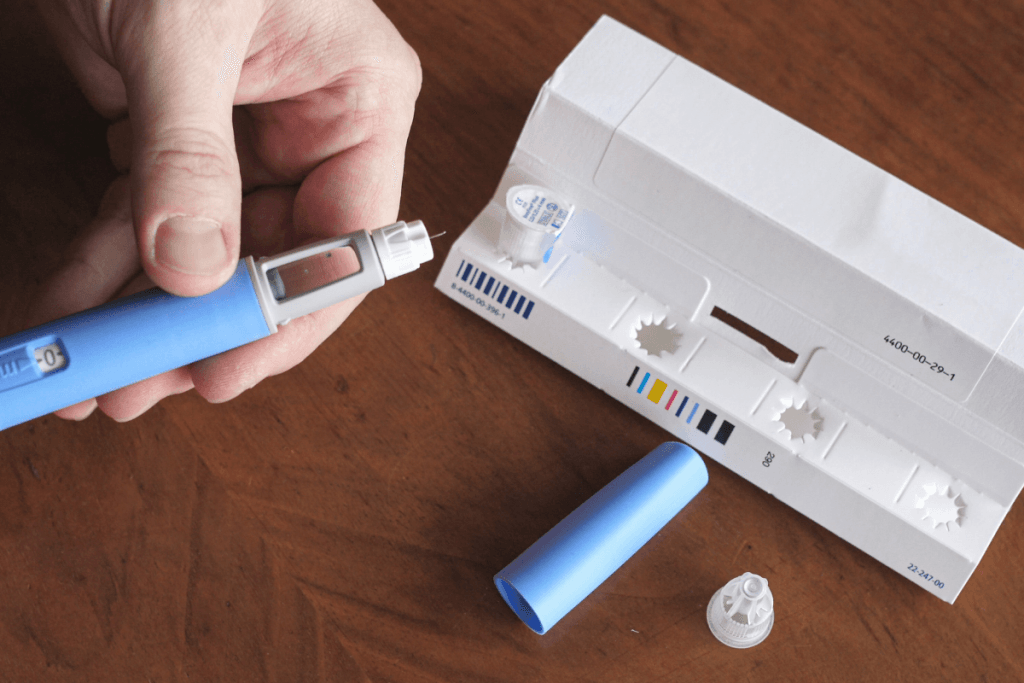
For clinic teams, does semaglutide need to be refrigerated is a practical inventory question. Storage affects potency, sterility risk, and waste. It also affects your ability to document quality. The answer depends on the specific semaglutide product, its container (pen versus vial), and whether it is manufacturer-made or compounded.
This page reviews label-driven storage expectations, what “left out” really means in practice, and how to think about dating terms like expiration date and beyond-use date (BUD; the discard date assigned to a compounded preparation). It is written for medication rooms, procedure clinics, and dispensing practices. MedWholesaleSupplies operates as a B2B supplier for licensed clinics and healthcare professionals, so the focus here stays on clinic workflows and compliance-ready documentation.
Core Concepts: Does Semaglutide Need To Be Refrigerated
Why refrigeration matters for injectable peptides
Semaglutide is a glucagon-like peptide-1 receptor agonist (GLP-1 RA; an incretin-mimicking medicine). Like many peptide-based injectables, it can degrade faster outside controlled storage. Heat, light, agitation, and repeated temperature cycling can increase chemical breakdown over time. In plain terms, prolonged warmth may reduce the medicine’s expected performance.
Refrigeration does not “improve” a product, but it slows degradation pathways. It also supports microbiological safety for products that are punctured or handled repeatedly. In clinics, this matters most at three points: receiving, storage on site, and after first use. Your standard operating procedures (SOPs) should define acceptable ranges, monitoring frequency, and escalation steps.
Brand-name injectable semaglutide: before first use versus in-use
Most clinic confusion starts when teams mix “unopened” storage with “in-use” storage. Manufacturer labeling often distinguishes these states. Unopened pens typically require refrigerated storage within a specified range. Once a pen is in use, some labels allow room-temperature storage for a limited period, with clear discard instructions. Those limits can differ across brands and presentations.
Note: “Room temperature” on labels usually has a defined maximum, not a vague concept. Clinics should align refrigerator logs and ambient temperature logs to the same units. When staff say a pen was “fine on the counter,” your documentation should capture actual time and conditions.
When you need additional context on product type, it can help to cross-reference clinic-facing summaries. For background on GLP-1 therapy in obesity care, see Wegovy Advancing Obesity Treatment for clinical context, and Ozempic For Weight Loss for terminology alignment in charts and consult notes.
Compounded preparations: stability and beyond-use dating
Compounded semaglutide storage is not the same problem as storage for an FDA-approved, manufacturer-sealed pen. A compounded preparation may be dispensed in a vial, may involve different excipients, and may carry a BUD that is based on USP standards, available stability data, and the compounding pharmacy’s process controls. That BUD is the controlling date for use in a clinic setting.
Clinics also see confusion around phrases like “compounded semaglutide shelf life” or “does compounded semaglutide need to be refrigerated.” The operational answer is: follow the compounding label instructions and retain the documentation. If the product arrives with a cold-chain requirement, treat it as required. If the label provides excursion allowances, document them exactly as written.
For practices that manage broader weight-management inventories, keep your storage guidance centralized. A condition hub can be useful for staff onboarding and cross-coverage. The Weight Loss page is one place to consolidate internal references and related items.
Temperature excursions: what “left out” can mean
A temperature excursion (time out of refrigeration) can be a brief transfer from receiving to the fridge. It can also mean a pen left in a staff bag, a vial in a warm medication room, or a product placed near a cooling vent that cycles. Each scenario changes risk. Duration, maximum temperature, container type, and whether it was previously opened all matter.
In incident reviews, teams often only record “left out overnight.” That phrase is not actionable on its own. Your log should specify: when it left controlled storage, when it returned, and the highest measured temperature if available. If the clinic cannot characterize the excursion, the safest operational stance is to quarantine and verify through the label or manufacturer resources.
Expiration date, in-use limits, and BUD are different controls
Clinics regularly see online claims like “does semaglutide expire after 28 days.” That “28-day” number is not a universal rule. It often reflects a specific in-use period for a specific product presentation, not the manufacturer’s printed expiration date. Some products allow a longer in-use window, while others allow a shorter one.
To reduce errors, treat these three terms as separate fields in your inventory system. The expiration date is assigned by the manufacturer for unopened product. The in-use limit is a discard timeline that begins after first use or after removal from refrigeration, depending on the label. The BUD applies to compounded preparations and depends on the compound, container, and sterility assumptions.
Practical Guidance
When staff ask, “does semaglutide need to be refrigerated,” your SOP should give a repeatable answer pathway. Start with product identification, then apply the relevant storage and dating rules. Standardize how you record events. A consistent workflow reduces both waste and compliance risk.
Use a simple intake process at receiving. Confirm the product name, NDC or identifier, lot number, and labeled storage requirements. Place it into the correct temperature zone promptly. If you run multiple GLP-1 products, label bins by product and “unopened/in-use” status to prevent mix-ups.
Tip: Build a one-page “excursion note” template for staff. It should capture who found the product, where it was stored, and estimated timelines.
Clinic checklist for storage control and documentation
- Assign ownership: One role reviews refrigerator logs daily.
- Track two dates: Expiration date and in-use/BUD date.
- Segregate inventory: Unopened stock separate from in-use items.
- Minimize handling: Avoid frequent moving in and out of refrigeration.
- Standardize labeling: Use consistent auxiliary labels for in-use dating.
| Scenario | Why it matters | What to document |
|---|---|---|
| Product arrived cool but not cold | May indicate partial excursion during transit or receiving | Arrival time, temp reading if available, quarantine decision |
| Semaglutide vial left out of fridge | Stability and sterility risk depends on time and container | Time out, max temperature, opened status, label guidance applied |
| Compounded semaglutide left out of fridge | BUD and storage limits may differ from brand labeling | Compounding label instructions, pharmacy contact, disposition note |
| Team unsure whether an item was opened | In-use limits may start at first use | Chain-of-custody review, lot trace, quarantine outcome |
For excursion decisions, avoid “best guesses.” If the label provides a room-temperature allowance, apply it exactly and record your rationale. If the label does not address the exact situation, escalate. That escalation could be to a pharmacist, your medical director, or manufacturer medical information, depending on governance.
Also separate operational handling from clinical decisions. Staff may notice reduced effect or increased adverse effects and wonder about “side effects of expired semaglutide.” Expired or improperly stored medication can be unpredictable, but clinics should not test it “to see.” Your quality system should define discard thresholds and reporting pathways for product complaints.
Compare & Related Topics
Because semaglutide sits in a broader GLP-1 landscape, teams sometimes generalize rules from one product to another. That is where “using semaglutide after 28 days” discussions often go off track. Some injectable GLP-1 products have different in-use windows, different temperature allowances, and different device designs. Always anchor decisions to the exact product labeling and your internal policy.
It also helps to separate brand-name semaglutide from other agents used in weight management. Liraglutide and tirzepatide have their own storage rules and device instructions. For related clinic reading, see Saxenda For Weight Loss for adjacent workflow considerations, and Mounjaro Weight Loss Insights for terminology when your formulary includes multiple injectables.
Another common question is “how long does semaglutide last in the body.” That pharmacokinetic topic is separate from storage, but it can influence patient counseling and missed-dose documentation. Prescribing information for semaglutide products describes a long half-life, which is one reason consistent handling and consistent administration matter at the program level. For program design context, Weight Loss Injections can help standardize language across your team.
When you need to reference example products in inventory systems, keep links and training materials specific. A product page like Ozempic Product can help staff confirm naming conventions, while Wegovy Flextouch can support device-specific differentiation in internal checklists.
Clinic Ordering and Compliance Notes
Inventory policy connects directly to sourcing and documentation. If a clinic is building a GLP-1 program, align purchasing, receiving, and storage SOPs before volume increases. Ordering through MedWholesaleSupplies is restricted to licensed clinics and credentialed healthcare professionals, which supports documentation-driven purchasing workflows and reduces mismatches between intended use and handling capacity.
In practical terms, does semaglutide need to be refrigerated becomes a compliance question once you add traceability. Your receiving record should retain lot numbers, supplier paperwork, and product identifiers in the same place as temperature logs. This makes audits and internal incident reviews faster. MedWholesaleSupplies supplies brand-name medical products through vetted distributors and verified channels, which helps clinics maintain clean provenance records for regulated inventory.
Plan storage capacity realistically. A small medication refrigerator that runs near full capacity tends to have more temperature swings and more door-open time. Consider dedicated bins for GLP-1 products, plus a clear quarantine area for excursions. If your practice spans weight-loss and body contouring services, keep categories separated to avoid handling errors. When you are mapping your catalog, Mounjaro Kwikpen can serve as an example of a different injectable class that still requires strict storage discipline.
Finally, set a policy for compounded products if your clinic uses them. Require that the compounding pharmacy label includes a BUD, storage conditions, and handling instructions. Retain certificates, invoices, and any provided stability rationale per your governance standards. If any element is missing, treat the product as nonconforming until clarified.
Authoritative Sources
For the most defensible answer to does semaglutide need to be refrigerated, start with the current prescribing information for the exact product you stock. Labels specify unopened storage ranges, in-use timelines, and defined temperature limits. They also describe when to discard after exposure to conditions outside the labeled range.
- FDA drug labels database (use to locate the latest Ozempic or Wegovy prescribing information).
- Ozempic prescribing information (Novo Nordisk) for storage, in-use dating, and excursion limits.
- Wegovy prescribing information (Novo Nordisk) for refrigerated storage requirements and room-temperature allowances.
In day-to-day clinic operations, treat labeling as the primary source. Use your internal SOPs to standardize documentation and escalation. When something does not fit the label, quarantine and verify before use. For deeper reading on GLP-1 program considerations, review Wegovy Advancing Obesity Treatment to keep team education consistent.
This content is for informational purposes only and is not a substitute for professional medical advice.