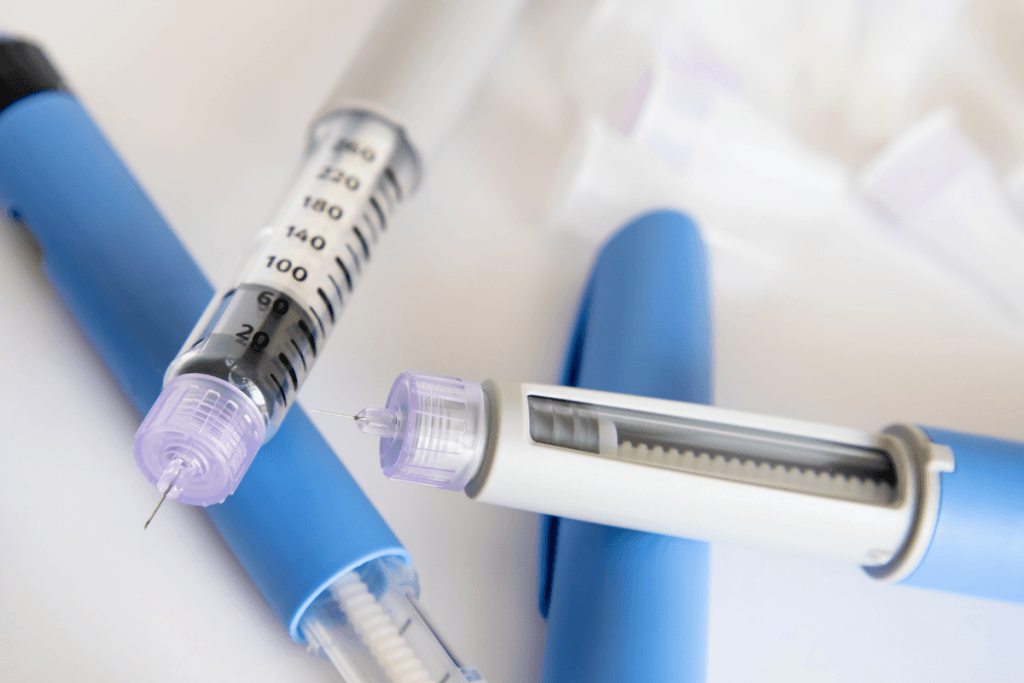
Ozempic for weight loss is a clinic workflow question as much as a clinical one. Ozempic is semaglutide, a GLP-1 receptor agonist (a medicine that mimics a gut hormone), but the exact brand, labeled indication, and patient context still drive how clinics screen, document, counsel, and follow up. In many settings, using Ozempic specifically for weight management may be off-label. That matters because documentation, informed consent, perioperative planning, adverse-event triage, and inventory controls all need to match the product actually being used.
Key Takeaways
- Product and indication matter because semaglutide brand names do not carry identical labeled uses.
- Off-label use raises charting, consent, and payer-facing documentation questions.
- Most early issues are gastrointestinal, but serious warnings still need a clear escalation script.
- Pen training, storage checks, lot tracking, and follow-up planning reduce workflow drift.
- Upcoming procedures should be reviewed early because GLP-1 therapy can affect anesthesia planning.
Ozempic for Weight Loss in Clinic Practice
Ozempic is an injectable semaglutide pen. For clinics, the first operational step is distinguishing the product from the broader class of weight-management therapies. A chart that only says semaglutide may be too vague for procurement, consent, and follow-up. Teams usually need the exact brand, the reason it is being discussed, prior GLP-1 exposure, and whether the case sits in diabetes care, obesity medicine, or both.
That distinction affects more than prescribing language. It can shape who performs intake, how informed consent is written, what prior records are requested, and whether the clinic needs a separate workflow for weight-management referrals. If your service line is still being formalized, the Clinic Operations hub can help frame intake, handoffs, and documentation standards across teams.
Why it matters: The product-indication distinction changes consent, charting, and perioperative communication.
Using a diabetes-labeled product for weight management may require clearer consent language and tighter chart notes. Clinics should make sure the documented indication, counseling points, and supplied product all align. Coverage rules, institutional policy, and referral expectations may also differ between obesity-labeled and diabetes-labeled products, even when the active ingredient is familiar to staff.
This page is written for licensed clinics and healthcare professionals.
Screening and Eligibility Factors
Not every referral is an operational fit for semaglutide-based care. When Ozempic for weight loss is being considered, the intake process should do more than capture weight and goals. It should establish indication, comorbidities, contraindications, concurrent medicines, and whether the clinic can support the monitoring burden that follows.
- Indication clarity: diabetes care, obesity management, or mixed referral reason.
- Relevant history: pancreatitis, gallbladder disease, severe GI symptoms, renal vulnerability, and pregnancy status.
- Boxed warning review: personal or family history relevant to medullary thyroid carcinoma or MEN 2.
- Concomitant therapy: insulin, sulfonylureas, or other drugs that may change risk.
- Eye and glycemic context: especially in patients with diabetes.
- Procedure planning: upcoming sedation, endoscopy, or surgery.
- Follow-up capacity: education time, reliable contact, and a defined adverse-event pathway.
Staff role design matters too. The clinician who decides appropriateness may not be the same person who trains on pen use, manages refill questions, or triages nausea calls. Clear role boundaries, competency records, and state-specific scope rules help clinics avoid drift; for team setup and delegated tasks, Clinic Team Requirements can support role-mapping discussions.
Baseline documentation does not need to be bloated, but it does need to be consistent. Clinics often capture the exact product under review, prior medication exposure, relevant history, current medicines, counseling completed, and the plan for follow-up. If use is off-label, the consent note should say so plainly and record that alternative options, known risks, and escalation steps were discussed.
Safety, Monitoring, and Counseling
Most tolerability issues are gastrointestinal, but the bigger operational risk is incomplete counseling or slow escalation. A structured Injection Safety approach helps teams separate expected early symptoms from events that need prompt review.
Common early issues
Early adverse effects can include nausea, vomiting, diarrhea, constipation, reduced appetite, and abdominal discomfort. Those complaints are common enough that clinics need a standard intake script for symptom calls rather than improvised advice. Staff should document onset, severity, fluid intake, ability to keep food down, and whether symptoms are improving, stable, or worsening.
- Nausea or early satiety: document onset, intake, and hydration tolerance.
- Vomiting or diarrhea: assess fluid loss and escalation thresholds.
- Constipation: review duration, severity, and associated pain.
- Abdominal discomfort: watch for persistence or worsening patterns.
- Injection questions: confirm site rotation and technique from current instructions.
These effects may be manageable, but repeated vomiting, poor oral intake, or symptom clusters that prevent hydration deserve closer review. Clinics should avoid normalizing every GI complaint simply because semaglutide is involved. The task is to decide which symptoms fit expected tolerability and which suggest a more serious problem.
Warnings that deserve a clear script
Counseling around Ozempic for weight loss should also cover label-based warnings and precautions. Teams commonly review the boxed warning related to thyroid C-cell tumor risk, hypersensitivity, pancreatitis, gallbladder problems, kidney injury that can follow dehydration, and the way concomitant diabetes therapies may change hypoglycemia risk. In patients with diabetes, vision changes or rapid glycemic shifts may warrant added attention.
- Persistent severe abdominal pain.
- Recurrent vomiting with dehydration signs.
- Right upper abdominal pain or jaundice.
- Widespread rash, facial swelling, or breathing difficulty.
- New neck mass, dysphagia, or persistent hoarseness.
Event handling should be scripted before the first prescription is written. Many clinics already apply that discipline in other injectable services, and the same logic behind Clinical Workflow Essentials translates well to semaglutide triage, documentation, and follow-up escalation. Serious or unexpected events may also require internal incident reporting or external reporting under local rules.
Operations: Pen Handling, Documentation, and Workflow
Safe use depends on a repeatable workflow, not a one-time conversation. Clinics should follow the current Instructions for Use for storage, preparation, injection, and disposal, while keeping internal logs for receiving, cold-chain exceptions, lot numbers, and expiry tracking. Counseling is incomplete if the clinic cannot also support pen training, traceability, and a clear path for symptom calls.
A simple clinic workflow
- Verify product and chart context: confirm the exact brand, indication, and current medication list.
- Review contraindications and precautions: include surgery plans, pregnancy considerations, and prior intolerance.
- Record consent and counseling: note off-label status when applicable and document warnings reviewed.
- Capture product traceability: log lot, expiry, dispense date, and any storage exception.
- Train on administration basics: use the current pen instructions, site rotation, and sharps disposal process.
- Schedule early follow-up: define who handles symptom calls, refill coordination, and missed visits.
- Log outcomes and events: update tolerance, adherence issues, and any adverse-event report.
The practical training piece is often underestimated. Patients may understand that semaglutide is self-injected and still confuse subcutaneous technique, site rotation, reuse practices, or disposal rules. Abdomen, thigh, and upper arm are common labeled subcutaneous sites, but the clinic should rely on the current product instructions rather than memory or copied handouts. Standardizing documentation across services can help; the planning logic used in broader Concepts and Workflow workups also reduces missed handoffs in medication clinics.
Ozempic for weight loss creates extra documentation pressure when the chart, consent form, and supplied product do not clearly align. That is where procurement, nursing, and prescriber workflows need the same version of the truth.
Brand-name inventory should come through verified supply channels.
Procedures and Anesthesia Planning
Pre-procedure screening is not optional for GLP-1 therapies. Semaglutide can delay gastric emptying (slower stomach emptying), which may matter before sedation, endoscopy, or anesthesia because aspiration risk assessment depends on whether the stomach may still contain food.
For clinic teams, the practical step is consistent intake and communication. Ask about current GLP-1 use, document recent administration according to your policy, and flag planned procedures early. Recommendations on holding therapy before surgery have evolved and may differ by patient risk, procedure type, and society guidance, so clinics should not rely on outdated scripts or one-size-fits-all timing rules.
Quick tip: Add recent GLP-1 use to every pre-procedure checklist.
If a patient reports persistent nausea, vomiting, bloating, or difficulty tolerating oral intake before a procedure, that information should reach the surgical or anesthesia team promptly. The goal is not to create blanket exclusions. It is to ensure the medication history is visible when airway and fasting plans are being made.
Where It Fits Among GLP-1 Options
Semaglutide-based workflows sit inside a larger medication category, and products are not interchangeable just because they share a class. For clinics, the key differences are labeled use, administration format, counseling burden, and how much retraining a switch creates.
| Product | Molecule or class | Typical labeled context | Clinic note |
|---|---|---|---|
| Ozempic | Semaglutide, GLP-1 receptor agonist | Type 2 diabetes management | Weight-management use may be off-label and should be documented clearly. |
| Wegovy FlexTouch | Semaglutide, GLP-1 receptor agonist | Chronic weight management | Similar class counseling does not remove the need for product-specific training. |
| Mounjaro KwikPen | Tirzepatide, GIP and GLP-1 agonist | Type 2 diabetes management | Do not assume the same label, pen workflow, or chart language. |
| Saxenda | Liraglutide, GLP-1 receptor agonist | Chronic weight management | Administration frequency changes teaching, follow-up, and adherence monitoring. |
A single master template with shared class counseling and product-specific fields is often easier to maintain. Many clinics already do this in other injectable services, using standardized contraindication and aftercare fields similar to broader Clinical Uses, Risks, and Workflow mapping.
In practice, Ozempic for weight loss belongs in a coordinated pathway that covers indication, consent, technique, monitoring, and perioperative review. The drug conversation and the operations conversation should be the same conversation.
Authoritative Sources
- Current FDA prescribing information for Ozempic injection
- American Society of Anesthesiologists guidance on GLP-1 medicines
- Official Ozempic pen use and storage instructions
Further reading for clinic teams usually starts with three checkpoints: confirm the exact labeled product, tighten consent language for the intended use, and keep perioperative screening current as guidance evolves.
This content is for informational purposes only and is not a substitute for professional medical advice.