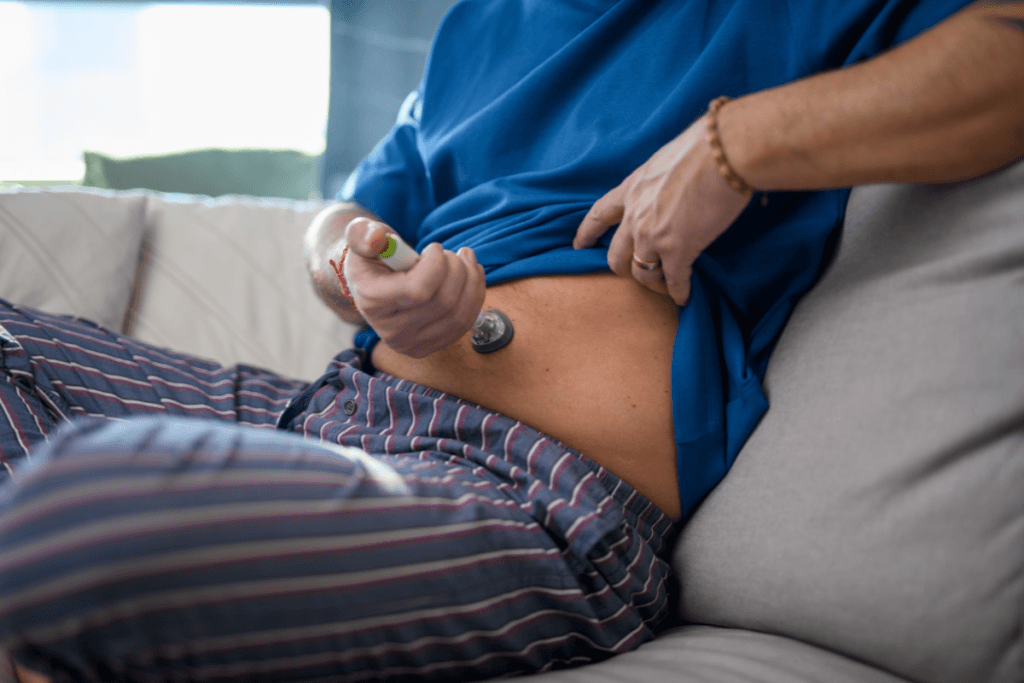
Mounjaro for weight loss is best viewed as a tirzepatide-based obesity-care question, not a simple product question. For clinicians, the value lies in how tirzepatide may reduce appetite, increase satiety (feeling full), and slow gastric emptying, while the risk lies in assuming every tirzepatide product carries the same indication, device, or monitoring needs. In practice, safe use depends on verifying the exact brand and label, screening for contraindications, and building follow-up around nutrition, activity, and tolerability.
Providers often use Mounjaro as shorthand for tirzepatide. That is understandable in conversation, but it can create avoidable errors in charting, procurement, and counseling. If you are reviewing stock, care pathways, or patient education materials, it helps to separate active ingredient, approved use, and clinic workflow before comparing options.
Key Takeaways
- Tirzepatide targets GIP and GLP-1 pathways.
- Brand name and approved use must be verified.
- Patient selection should include risk and follow-up capacity.
- Monitoring should focus on GI tolerance and escalation triggers.
- Wraparound obesity care still matters for long-term fit.
Mounjaro for Weight Loss in Obesity Care
In obesity care, Mounjaro is relevant because it contains tirzepatide, but brand recognition should not replace indication review. The listing for Mounjaro KwikPen is useful for identifying a specific presentation, while the broader Weight Loss Hub helps teams distinguish educational content from item-level listings. For clinic workflows, that separation reduces documentation errors and keeps procurement discussions grounded in the exact product being sourced.
Why it matters: Product name, active ingredient, and approved use are not always interchangeable.
In the US, Mounjaro carries a type 2 diabetes indication, while tirzepatide also has separate branded weight-management labeling. Other markets may package or describe products differently. For procurement and clinical governance, the practical rule is simple: verify the exact branded product, the official labeling in your market, and the intended use before it enters a patient-facing pathway. That check is especially important when staff shorthand uses one brand name to describe an entire drug class.
Obesity care also benefits from a wider lens. Tirzepatide is not a stand-alone answer to a chronic, relapsing condition. It fits best within documented care plans that address nutrition, physical activity, sleep, behavioral support, and cardiometabolic comorbidity review. When teams set expectations this way, follow-up conversations tend to be clearer and less reactive.
Access is intended for licensed clinics and healthcare professionals.
How Tirzepatide Drives Weight Reduction
Tirzepatide drives weight reduction through dual incretin activity. It targets both GIP and GLP-1 pathways, which can lower hunger, enhance fullness after meals, and slow how quickly the stomach empties. The combined effect may reduce total energy intake over time. In patients who also have type 2 diabetes, glycemic improvement and weight change may occur together, which is why medication review at baseline matters.
Mechanism in practical terms
In day-to-day care, the mechanism often shows up as earlier satiety, smaller portion tolerance, and less drive to snack between meals. The same biology can also explain why gastrointestinal (digestive) symptoms are common early on. Nausea, vomiting, diarrhea, constipation, and reduced appetite are part of the tolerability picture, not separate unrelated events. Teams that explain this upfront usually document better adherence conversations.
Delayed gastric emptying is another clinically useful concept. It can influence meal size, but it also matters when patients use other oral medicines or are preparing for procedures that involve sedation or anesthesia. For class context, some clinics compare tirzepatide with GLP-1 products such as Ozempic or Wegovy FlexTouch 1 mg. Those comparisons are helpful, but they should focus on mechanism, label context, and workflow fit rather than assuming the products are interchangeable.
Expectation setting also matters. Mounjaro for weight loss should be framed as a gradual intervention measured over routine follow-up, not by day-to-day scale noise. Early visits are often more useful for tolerability, hydration, injection confidence, and care-plan reinforcement than for judging the final response trajectory.
Products are sourced through vetted distributors.
Where It Fits in Patient Selection and Care Planning
Mounjaro for weight loss is most useful as a structured selection question: who is likely to benefit, who needs extra caution, and what support will the clinic need to provide? A sound starting point includes obesity severity, obesity-related complications, diabetes status, prior exposure to incretin therapies, gastrointestinal history, and the clinic’s ability to monitor response over time. Administrative fit matters too. If follow-up is fragmented, even a strong medication option can create poor documentation and avoidable discontinuation.
Clinical factors to review before initiation
- Indication match: Confirm the exact brand and approved use in your market.
- Baseline status: Document weight, comorbidities, and current metabolic therapy.
- History review: Screen for pancreatitis, gallbladder issues, or severe GI intolerance.
- Warning checks: Review thyroid tumor history and MEN2 risk where labeled.
- Medication overlap: Note insulin or secretagogue use before follow-up planning.
- Care capacity: Make sure injection training and symptom review can be supported.
Patient counseling should stay practical. Explain that appetite suppression may reduce meal size, but nutrition quality, protein adequacy, hydration, sleep, and physical activity still need active support. If your team compares adjacent product pathways, a page such as Saxenda can help illustrate that not all injectable weight-management agents share the same schedule, device, or evidence base. The broader Weight Loss Products page can also help procurement teams separate category planning from a single product discussion.
One useful counseling frame is to describe tirzepatide as a facilitator, not a replacement, for behavior change. That avoids the false choice between medication and lifestyle support. In practice, wraparound care is often what makes weight-loss expectations more realistic and monitoring more consistent across primary care, obesity medicine, and specialist referral pathways.
Referral thresholds should also be explicit. Endocrinology, obesity medicine, or bariatric referral may be reasonable when weight complexity is high, comorbid disease is difficult to coordinate, or prior medication intolerance narrows choices. A provider-facing tirzepatide pathway works best when referral rules are written down before a patient has a problem, not after.
Safety, Contraindications, and Monitoring
The key safety questions usually involve contraindications, gastrointestinal tolerability, metabolic overlap, and escalation triggers. Before starting tirzepatide, clinics should review current official labeling for boxed warnings and contraindications, especially where there is a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2. The practical point is not to memorize every line item from memory, but to make label review part of the workflow.
Common adverse effects are usually gastrointestinal. Nausea, vomiting, diarrhea, constipation, dyspepsia, and reduced appetite may appear early or after treatment changes. These effects may be manageable for some patients, but persistent vomiting, poor oral intake, or volume depletion deserve closer attention because they can complicate hydration status and overall tolerance. When tirzepatide is used alongside insulin or insulin secretagogues, the team also needs a plan to watch for hypoglycemia within the relevant prescribing framework.
Another overlooked issue is procedure planning. Because gastric emptying can be delayed, clinics should reconcile current peri-procedural guidance before sedation or anesthesia. That is especially important when obesity treatment is being coordinated across primary care, surgery, gastroenterology, or procedural services.
Monitoring signals that deserve closer review
- Persistent vomiting or poor intake
- New severe abdominal pain
- Possible gallbladder symptoms
- Unexpected hypoglycemia episodes
- Dehydration or orthostatic symptoms
- Injection-use confusion or missed follow-up
Mounjaro safety and monitoring should also include documentation quality. Record the product used, the clinical reason for treatment, baseline risk factors, counseling provided, and any symptom escalation plan. This makes later tolerance decisions more defensible and reduces ambiguity when multiple teams touch the same chart.
Monitoring is not just about side-effect capture. It also includes confirming that weight-management goals, cardiometabolic priorities, and treatment burden still line up. If a patient is skipping meals, developing persistent aversion to intake, or struggling with the injection routine, the issue may be care-plan fit rather than simple nonadherence. That distinction matters when documentation is reviewed later.
Clinic Workflow and Sourcing Checks
For provider teams, the operational side of tirzepatide matters almost as much as the mechanism. Product verification, documentation, storage, and device familiarity all affect whether care stays safe and consistent. Clinics should confirm the active ingredient, brand name, presentation, lot, expiry, storage instructions, and label context before any item moves from procurement to dispensing or administration workflow.
Quick tip: Separate brand shorthand from active-ingredient review during intake, stock control, and charting.
- Confirm brand-label alignment
- Verify clinic purchasing status
- Match device to protocol
- Record lot and expiry
- Follow labeled storage steps
- Document counseling and follow-up
If your team stocks multiple injectables, keep look-alike and sound-alike risks in mind. Weekly products can still differ in indication, pen design, and patient education needs. Reviewing a specific listing such as Saxenda Pens can be a useful reminder that device handling and administration cadence are not uniform across the category. Policies for storage and handling should always follow the official product labeling and your clinic’s internal controls.
Supply channels are verified before products reach licensed clinics.
For sourcing, governance matters. Separate procurement approval from clinical approval, keep receiving records consistent, and make sure staff know where current label documents are stored. This is especially helpful when obesity care intersects with diabetes care, procedural services, or satellite clinic locations that may not all use the same formulary language.
Device education should be standardized across staff. Even experienced teams benefit from a short script that covers injection-day logistics, storage handoffs, and when to contact the prescribing team. Standardization reduces the variability that often appears when obesity care is added onto an already busy primary care or multispecialty workflow.
How Tirzepatide Compares With Other Injectable Options
The main comparison point is not whether one brand is familiar. It is how mechanism, label, administration cadence, and monitoring demands fit the clinical setting. Tirzepatide is a dual GIP and GLP-1 receptor agonist, while semaglutide and liraglutide are GLP-1 receptor agonists. That difference helps explain why Mounjaro for weight loss often enters discussions about efficacy expectations, but practical selection still depends on individual risk review and the exact product being used.
| Approach | Receptor Target | Typical Context | Operational Note |
|---|---|---|---|
| Tirzepatide products | GIP and GLP-1 | Context depends on exact brand and label | Verify indication, device, and warning language |
| Semaglutide products | GLP-1 | Diabetes or weight management depending on product | Do not assume interchangeable labeling |
| Liraglutide products | GLP-1 | Weight management or diabetes, product dependent | Cadence and training needs may differ |
No injectable option removes the need for wraparound care. Clinics still need protocols for baseline review, symptom monitoring, nutrition support, follow-up intervals, and escalation when red-flag symptoms appear. When teams compare products this way, the conversation becomes less about hype and more about operational fit.
From a procurement angle, comparison should also include stocking complexity. The more pens, labels, and education leaflets a clinic carries, the greater the need for clear segregation and version control. That is one reason many services build a small, deliberate protocol set rather than treating every incretin product as a routine substitute for another.
Further reading should follow the same logic: use category hubs for browsing, product pages for presentation details, and official labels for contraindications, warnings, and approved uses.
Authoritative Sources
- For boxed warnings and contraindications, review the Mounjaro prescribing information.
- For US weight-management approval context, see the FDA announcement on tirzepatide for chronic weight management.
- For wraparound care expectations in practice, refer to NHS England guidance on tirzepatide in primary care.
This content is for informational purposes only and is not a substitute for professional medical advice.