Orthovisc and Synvisc are both viscosupplementation options used in knee osteoarthritis care, but they are not the same product. This Orthovisc vs Synvisc: Clinic-Facing Comparison Guide focuses on the differences that usually matter in practice: formulation and source, avian-exposure screening, product-family scheduling, post-injection monitoring, and payer documentation. For most clinics, the useful question is not which brand wins in the abstract. It is which option fits the chart, the workflow, and the current coverage rules.
Key Takeaways
- Both products are hyaluronan-based knee injection options, but their formulations differ.
- Source material can matter for allergy review and product selection workflows.
- Brand families and product variants may affect scheduling and authorization steps.
- Coverage decisions usually depend on policy criteria and documentation, not brand name alone.
- Teams should document diagnosis, prior therapy, lot details, and post-injection reactions carefully.
The intended audience here is clinic teams and licensed medical professionals.
Orthovisc vs Synvisc at a glance
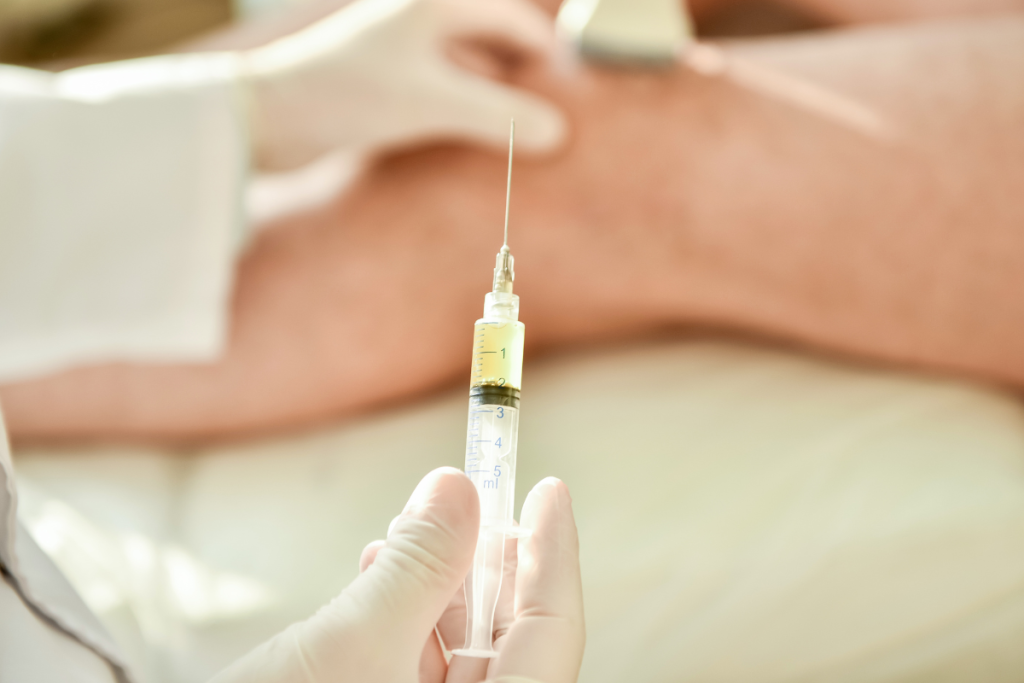
The short clinical answer is that Orthovisc and Synvisc are both hyaluronan-based knee injections, often called gel injections for knees, but they differ in formulation. Orthovisc is generally described as a sodium hyaluronate product with non-avian sourcing, while Synvisc contains hylan G-F 20 and is classically avian-derived. That distinction can matter when clinics review allergy history, stock product, or explain why two hyaluronic acid products are not automatically interchangeable.
It also helps to separate class questions from brand questions. Viscosupplementation evidence is mixed, and major guidance has not been uniform about routine use in knee OA. That means Orthovisc vs Synvisc decisions often come down to operational fit, prior response, and policy language rather than a clear, across-the-board efficacy winner. In a busy practice, those differences affect far more than the injection room. They shape ordering, scheduling, documentation, and follow-up.
| Dimension | Orthovisc | Synvisc | Why Clinics Care |
|---|---|---|---|
| Base material | Sodium hyaluronate | Hylan G-F 20 | Different label wording and chart terminology |
| Source context | Commonly described as non-avian | Commonly described as avian-derived | Relevant when reviewing avian sensitivity history |
| Product-family planning | Verify the exact item and current labeled regimen | Verify the exact Synvisc variant and current labeled regimen | Visit count and authorization wording may differ |
| Reaction review | Local joint reactions can occur | Local joint reactions can occur | Supports counseling and follow-up planning |
| Coverage workflow | Policy criteria still apply | Policy criteria still apply | Most denials relate to documentation or policy fit |
Why it matters: Source and product-family details can change screening, scheduling, and claim submission.
How to interpret effectiveness claims
The safest clinic-facing answer is that no single product can be called the most effective knee gel injection for all patients. Published comparisons vary in design, endpoints, retreatment windows, and product families. Some analyses suggest similar overall class performance, while individual studies may favor one product under certain conditions. That is not strong enough to treat any brand as the default winner across every orthopedic, rheumatology, or sports medicine workflow.
Another reason comparison headlines can mislead is that studies do not always measure the same thing. Pain scores, function scores, rescue medication use, timing of follow-up, and repeat-treatment criteria may differ. A product that appears stronger in one design may not translate cleanly into a stocking rule for every service line. Clinics usually get more value from comparing chart fit, prior response, scheduling burden, and coverage requirements than from chasing a single headline result.
Professional guidelines also shape how teams interpret benefit. Some groups have been cautious about routine viscosupplementation use in knee OA because observed benefit may be modest or inconsistent across populations. That does not make every use inappropriate. It does mean clinics should read effectiveness claims beside the current label, the broader guideline environment, and the practice’s own protocol instead of assuming a universal hierarchy.
The comparison points that usually drive clinic decisions
The most useful Orthovisc vs Synvisc comparison is usually operational. Both sit in the same treatment category, but formulation, naming, and variant differences can create downstream problems if the ordered product, authorized product, and stocked product do not match.
Formulation and source review
Orthovisc is typically discussed as a sodium hyaluronate product produced through bacterial fermentation, while Synvisc is a hylan product derived from avian material. Clinics do not need to treat that distinction as shorthand for better or worse performance. They do need to treat it as a real labeling and screening point. If a chart mentions sensitivity to avian proteins, feathers, or egg products, teams usually want the product source reviewed before the visit, not at the chairside.
This point also matters when procurement and clinical staff are different people. The ordering team may think in brand names, while the injecting clinician may think in class terms such as hyaluronic acid knee injections. A clean workflow links both. The order, prior authorization, and chart note should identify the exact item and the reason it was selected.
Product variant and visit planning
Brand families in this category can include differently named variants and different scheduling expectations. That affects reminder calls, room turnover, consent forms, and how staff explain the treatment plan. Clinics should verify the exact product variant and current labeled regimen each time rather than rely on memory or assume all products in the same class use the same pattern.
This is one reason brand comparisons can feel simple on paper and messy in practice. A single name in the chart is not always enough. Teams often need the full product name, the current labeling, and the intended visit sequence aligned before the first injection is scheduled. That is especially important when more than one viscosupplement or more than one knee injectable is stocked at the same site.
Inventory and labeling discipline
Operational errors with viscosupplementation often start before the patient arrives. Common failure points include mismatched product names on authorization requests, unclear charge descriptions, and missing lot documentation after administration. Storage and handling should follow the current label and supplier instructions, not assumptions based on another injectable in the practice.
When more than one musculoskeletal injectable is stocked, simple standardization helps. Use a consistent naming convention, keep the exact product variant visible in the EHR, and make lot and expiration capture part of the routine procedure note. Those small steps reduce rework when claims are reviewed later or when the team needs to assess a post-injection event.
Safety, reactions, and cautions
The main safety issues are usually local, not systemic. Clinics most often monitor for post-injection pain, swelling, warmth, stiffness, or effusion (fluid buildup) in the treated knee. These reactions may be mild and self-limited, but they still matter because they affect follow-up calls, patient instructions, and the decision to reevaluate quickly.
Viscosupplementation products can also trigger an acute inflammatory response that resembles infection. That is why documentation matters. Time from injection, degree of swelling, ability to bear weight, skin findings, and fever history help frame the next step. If symptoms are marked or progressive, the differential should stay broad until septic arthritis and other urgent causes are excluded.
Many safety discussions blend class-wide effects with product-specific cautions. Synvisc’s avian-derived origin may be relevant when the medical record includes avian protein, feather, or egg sensitivity. Orthovisc’s non-avian sourcing may be relevant when clinics are trying to avoid that exposure context. Even so, brand choice should still follow the current label, the full allergy history, and the treating clinician’s judgment. Source alone does not answer every safety question.
- Review infection risk before injection.
- Document baseline pain and swelling.
- Record aspiration details when performed.
- Capture lot, site, and laterality.
- Escalate marked swelling or fever promptly.
Clear post-procedure documentation also helps with repeat-treatment discussions. A prior note that simply says tolerated well may be too thin to support later decision-making. A better note describes immediate response, any flare, and whether further evaluation was needed.
Coverage, prior authorization, and Medicare questions
Coverage is usually driven by payer rules and documentation, not by a simple Orthovisc vs Synvisc ranking. Many plans evaluate hyaluronic acid knee injections against criteria such as symptomatic knee OA, prior conservative therapy, timing of previous injections, and the exact product requested. A denial often reflects missing or mismatched documentation more than the brand itself.
This is why teams sometimes ask why Orthovisc is not covered or whether Medicare will pay for a specific viscosupplement. The practical answer is that coverage can vary by commercial plan, Medicare Advantage plan, and Medicare Administrative Contractor. Some policies are product-specific. Others address the class more broadly. Either way, clinics need the current policy language before they schedule or reorder.
Common reasons a request stalls
- Exact product variant not identified.
- Diagnosis or laterality is unclear.
- Conservative therapy history is thin.
- Repeat-treatment timing is not supported.
- Prior response is not documented.
- Policy rules changed since the last request.
For payer review, the cleanest chart usually includes the diagnosis, symptom burden, prior measures already tried, prior injection history when relevant, and the exact product or variant requested. If your team bills from a hospital outpatient department, physician office, or ambulatory surgery setting, make sure the operational pathway matches the place-of-service rules that apply there.
Traditional Medicare often depends on local contractor guidance rather than one universal national rule for every hyaluronic acid product. That is why a protocol that worked in one state or one year may not transfer cleanly to another setting. Build periodic policy checks into the workflow instead of treating prior approval history as permanent.
Quick tip: Confirm the exact product name before submitting prior authorization.
Product sourcing references reflect vetted distributors and verified supply channels.
A practical clinic workflow before ordering or administering
A consistent workflow prevents more problems than a brand debate. For most practices, the key is to connect procurement, scheduling, documentation, and follow-up so the same product details appear at every step.
- Confirm diagnosis and laterality.
- Review allergy and prior reaction history.
- Match the requested brand and variant.
- Check current payer policy and authorization status.
- Verify label-based storage and handling steps.
- Prepare lot, expiration, and site documentation fields.
- Record response and any early adverse events.
This kind of checklist is especially useful when different staff members order, receive, and administer the product. It reduces rework, helps with audits, and supports cleaner follow-up if a repeat course is later considered or a post-procedure question arises.
Where they fit among knee osteoarthritis injection options
Neither product is clearly the most effective knee gel injection for every patient or every clinic. Evidence across viscosupplementation products is heterogeneous, and head-to-head comparisons do not provide a simple universal winner. In practice, clinics usually compare prior response, safety context, visit burden, coverage, and formulary fit rather than chase a single best label.
That broader view matters because viscosupplementation sits beside other knee OA approaches, not above them. Physical therapy, weight management, bracing, oral or topical agents, corticosteroid injections, and surgical referral pathways may all enter the discussion depending on the case and the service line. A clinic-facing comparison should therefore ask where a product fits in care, not only how it compares on a marketing grid.
From an operations standpoint, standardizing one or two clearly defined pathways may be more useful than stocking many overlapping options. That does not mean every clinic should make the same choice. It means the decision should be anchored to labeled use, workflow complexity, prior authorization burden, and the type of follow-up the practice can support.
If you are reviewing related stocking or workflow topics across musculoskeletal injectables, the Joint Injections hub provides broader category context. It can help teams compare how different injection types affect documentation, handling, and service-line planning.
One more practical point: do not assume that a well-known brand name solves patient selection or follow-up questions. The value of any viscosupplement workflow depends on clear indications, consistent documentation, and realistic expectations about variable response.
Authoritative Sources
For label details and broader clinical context, start with these references:
- AAOS guideline on knee osteoarthritis management
- Current DailyMed labeling search for ORTHOVISC
- Current DailyMed labeling search for Synvisc
In short, the difference between Orthovisc and Synvisc usually comes down to formulation, source context, product variant, documentation burden, and payer fit. Compare the current label, the clinic workflow, and the policy language together before standardizing a protocol.
This content is for informational purposes only and is not a substitute for professional medical advice.