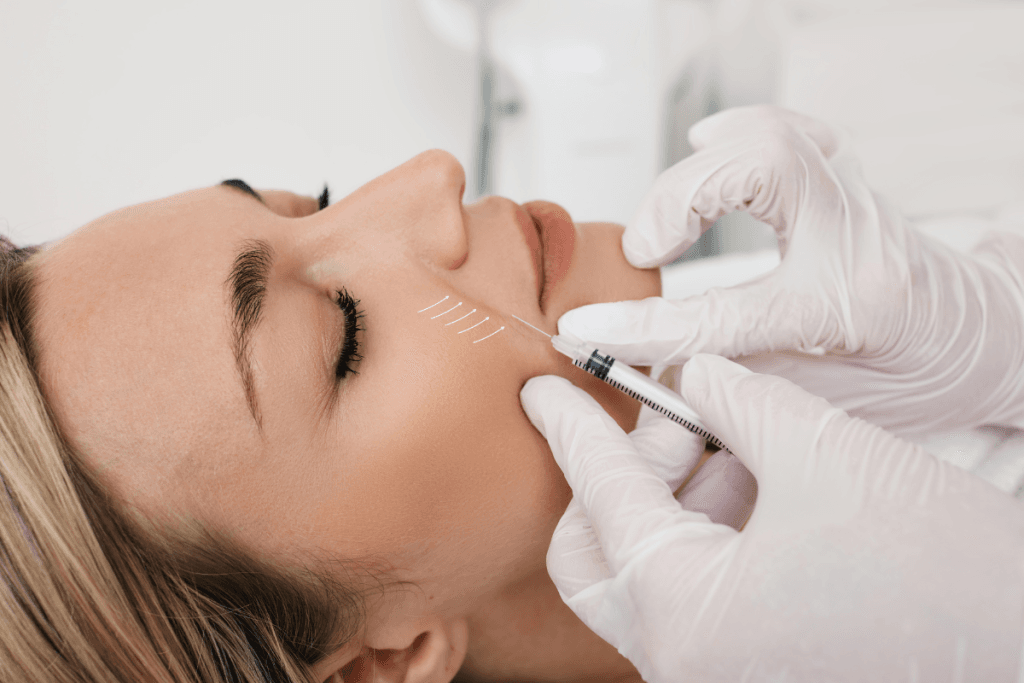
Ultracol injection for skin rejuvenation is generally evaluated as a polydioxanone (PDO) biostimulator used to support gradual collagen remodeling and skin-quality improvement. For licensed aesthetic clinics, the practical questions are not only what it is, but where it fits, who may suit it, how long changes may take to appear, and what safety and workflow controls need to be in place before use. That matters because a collagen-stimulating injectable behaves differently from a hydration-focused skin booster, a volumizing filler, or a thread-based lift.
Key Takeaways
- PDO-based category: collagen support is the core idea, not instant bulk volume.
- Best-fit goals: texture, firmness, and mild laxity rather than dramatic lifting.
- Timeline matters: meaningful change is usually assessed over repeat follow-up visits.
- Workflow counts: verify labeling, treatment areas, handling, and traceable documentation.
Ultracol Injection for Skin Rejuvenation in Context
Ultracol is generally described as an injectable PDO-based biostimulator, or collagen-stimulating injectable. In plain language, that means clinics are usually considering it for gradual improvement in skin quality rather than for immediate contour creation. The discussion tends to center on firmness, texture, early laxity (skin looseness), and broader rejuvenation planning.
That positioning places it between categories. It is not mainly a classic hyaluronic acid volumizer, and it is not the same as a suspension thread or surgical lift. The first planning question is simple: are you trying to improve skin quality over time, or are you trying to create immediate shape, projection, or repositioning? If the goal is glow-first or hydration-first care, the broader Skin Boosters Category and Skin Boosters Product Hub help frame the wider landscape.
Common clinic conversations around this category include facial rejuvenation, fine lines, elasticity support, and mild textural change in skin-quality treatment zones. Exact indications, preparation steps, and approved treatment areas may vary by market, so clinics should rely on current product documentation rather than informal summaries, social posts, or shorthand naming.
Because this is a gradual-change category, expectation setting matters early. A patient asking for a visible same-day lift or significant volume correction is often asking for a different mechanism. A patient willing to accept staged improvement, reassessment, and photo-based follow-up may be a better fit for a biostimulator pathway.
MedWholesaleSupplies serves licensed clinics and healthcare professionals.
Why it matters: A collagen-stimulating injectable needs different consent language than an instant-volume filler.
Who May Be a Good Candidate and Who Needs Caution
Best-fit candidates usually want progressive improvement in texture, firmness, and mild laxity rather than a dramatic one-visit transformation. In practice, clinics often find this category more useful for patients with early or moderate skin-quality concerns, reasonable expectations, and willingness to return for planned review.
Common Candidate Profiles
- Early laxity concerns: prefers gradual tightening over immediate reshaping.
- Texture-focused complaints: prioritizes skin quality more than projection.
- Maintenance mindset: accepts staged assessment and baseline photography.
- Combination plans: may pair with surface or hydration-focused treatments.
- Realistic expectations: understands collagen remodeling is progressive.
Pause Points Before Treatment
- Active infection: postpone until the skin is clinically clear.
- Unclear indication: confirm the target area matches current labeling.
- Immediate volume request: another class may fit the goal better.
- Recent procedures: spacing may matter after resurfacing or energy devices.
- Documentation gaps: resolve consent, history, and product checks first.
Screening should also address prior injectable history, prior biostimulators or fillers in the same area, inflammatory skin conditions, bleeding risk, relevant systemic history, and known hypersensitivity to listed ingredients. Not every rejuvenation complaint is a match. Deeper folds, more advanced ptosis, or patients seeking obvious structural lift may require a different approach or a combination plan.
For many practices, Ultracol injection for skin rejuvenation fits better when the plan emphasizes staged review, baseline photography, and gradual change. When the case is really about hydration or broader class selection, references such as Restylane Skinboosters and Types Of Dermal Fillers can sharpen the conversation without forcing a poor match.
How It Works, When Results May Appear, and Maintenance
Results are usually gradual because collagen remodeling takes time.
Ultracol is positioned as a collagen-stimulating injectable rather than a simple hydrating product. After placement, the PDO-based material may act as a stimulus for tissue remodeling, so clinics are typically looking for progressive change in texture, firmness, or mild laxity rather than a dramatic same-day transformation. This also explains why it should be judged against skin-quality endpoints, not against bold lifting claims.
Some patients may notice early changes from post-procedure edema, tissue support, or surface hydration, but the clinically meaningful effect is usually judged over follow-up visits, not in the first few days. Standardized photography, consistent lighting, and repeat assessment points matter more than anecdotal day-one feedback. In clinic communication, that means setting expectations around progression rather than promising a fixed result window.
Maintenance can be part of some biostimulator treatment plans, but there is no single schedule that fits every practice. The need for repeat treatment depends on current labeling, local regulation, anatomical area, baseline skin quality, and how the clinic defines a successful endpoint. If a market lists multiple configurations or product codes, document them separately rather than assuming the same protocol or outcome profile applies across all versions.
Area choice also matters. Facial zones, neck rejuvenation, and other skin-quality indications may not behave the same way, and not every market will list the same treatment areas. Clinics should confirm current instructions for use before applying one area-specific workflow to another.
Safety Considerations and Follow-Up
The main safety issues are the familiar ones seen across facial injectables: technique-related complications, bruising, swelling, infection risk, delayed inflammatory reactions, nodules, contour irregularity, and mismatched expectations.
Screening should cover prior filler or biostimulator history, active skin infection, recent resurfacing or energy-based treatment, inflammatory dermatoses, bleeding risk, relevant autoimmune history when appropriate, and any known hypersensitivity to listed ingredients. Clinics should also confirm approved treatment areas, contraindications, and preparation steps from the current instructions for use, because those details may vary by market.
Product choice does not replace anatomy. Safe injectable practice still depends on sound assessment, correct plane selection, careful technique, aseptic handling, and a documented escalation pathway. Clinics evaluating any biostimulator should be prepared for the same operational questions they use with other advanced injectables: what is the expected short-term recovery pattern, what symptoms need same-day review, and what complication response pathway is already in place?
Expected Reactions vs Red Flags
- Common short-term changes: tenderness, swelling, bruising, and temporary unevenness.
- Injection-site lumps: may reflect placement, edema, or early tissue reaction.
- Escalate same day: severe pain, blanching, dusky color change, or livedo.
- Infection concern: fever, drainage, increasing warmth, or worsening redness.
- Delayed review: persistent nodules or inflammation outside the expected course.
Aftercare should be simple, written, and product-specific. Many clinics avoid stacking additional procedures too quickly if that could confuse swelling or tenderness, and they provide a clear route for same-day review if symptoms escalate. A standardized follow-up pathway matters as much as the injection itself, especially when the intended result is gradual and may be confused with early post-procedure change.
Clinicians should also keep an escalation plan for suspected vascular compromise, infection, or delayed inflammatory response. Even when the likelihood is low, rapid recognition and documented response are part of safe injectable practice. If your team uses consistent follow-up templates across aesthetic treatments, keep the language simple, specific, and tied to symptom thresholds rather than vague reassurance.
How It Compares With Skin Boosters, Fillers, and Threads
Ultracol sits closer to a collagen-support strategy than to an instant volumizer.
| Approach | Main aim | What clinics usually expect | Key operational question |
|---|---|---|---|
| PDO biostimulator | Gradual skin-quality support | Progressive textural and firmness change | Is the goal collagen support rather than instant shape? |
| HA skin booster | Hydration and surface quality | Glow, hydration, and fine textural refinement | Is water-binding improvement the main priority? |
| PLLA or CaHA category | Biostimulation with differing material behavior | Structural support or later remodeling, depending on class | Which mechanism best fits the tissue target and workflow? |
| Thread lift | Mechanical lift and repositioning | Immediate lift component with a different risk profile | Is suspension the real treatment goal? |
In day-to-day planning, clinics often compare PDO biostimulators with hydration-led injectables, PLLA or CaHA categories, and thread-based lifting. The right choice depends on the primary endpoint: glow and hydration, gradual collagen support, structural contour, or mechanical lift. A thread lift answers a different question than a collagen-stimulating injectable, so the two should not be treated as direct substitutes.
For adjacent category context, compare options such as Profhilo Structura, Rejuran Healer, and Radiesse 3 mL. These are not interchangeable products, but reviewing them side by side helps clinics think in mechanisms rather than hype. That is often more useful than asking which option is simply “best.”
Clinic Workflow: Verification, Documentation, and Handling
Before a clinic adds any new biostimulator, the operational questions matter as much as the science.
Before adopting Ultracol injection for skin rejuvenation, confirm the exact product identity, packaging, market authorization, and training expectations for your jurisdiction. Avoid relying on shorthand names, informal social posts, or assumptions drawn from a different PDO product, especially when multiple configurations may exist across markets.
Lot traceability, shelf-life review, and documented receipt conditions should be part of the same chain-of-custody process you use for other advanced injectables. If the labeling, packaging, or source documentation is unclear, pause the workflow until the information is verified.
Pre-Treatment File Checklist
- Verify exact SKU: record product name, batch, and expiry.
- Confirm label status: indications, areas, and current IFU version.
- Screen the patient: history, expectations, and recent procedures.
- Capture baseline data: consent, photos, and treatment rationale.
- Plan the session: area mapping and follow-up timing.
- Record handling steps: storage conditions and preparation notes.
- Define escalation route: same-day review and complication contacts.
Products are sourced through vetted distributors and verified supply channels.
If your team standardizes intake and follow-up across services, related references on Mesotherapy Workflow, PRX-T33 Basics, and Dysport Aftercare can help align documentation habits, consent language, and post-treatment review steps.
Quick tip: Record the exact product name, batch, and expiry at the same visit.
Storage, preparation, handling, and disposal should always follow the current instructions for use. If any detail is unclear, stop and verify it with the manufacturer documentation or your authorized distributor rather than inferring from a similar injectable. Clinics that treat product verification as a routine step usually reduce avoidable confusion later in the patient file.
Authoritative Sources
- For safe injection standards, CDC Injection Safety.
- For general cosmetic injectable risk context, FDA Dermal Fillers and Soft Tissue Fillers.
- For a peer-reviewed regenerative case paper, NIH/PMC Regenerative Synergy in Facial Rejuvenation.
In short, Ultracol is best assessed as a gradual collagen-supporting option for skin quality rather than a shortcut to instant lift or full-volume correction. The strongest clinic decisions come from careful screening, realistic timeline counseling, sound technique, and disciplined documentation.
This content is for informational purposes only and is not a substitute for professional medical advice.