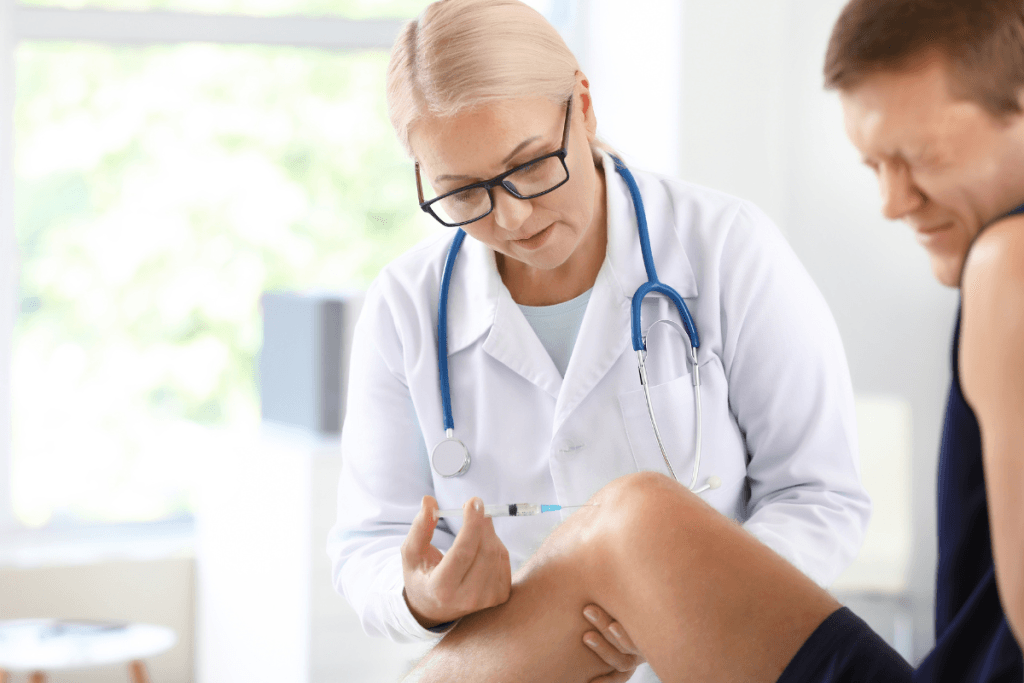
Hyalgan vs Euflexxa is usually not a question of naming one universal winner for knee osteoarthritis. Both are intra-articular (inside-the-joint) hyaluronic acid viscosupplements used in knee OA programs, but clinics usually separate them by workflow fit rather than simple brand preference. The practical differences are the treatment series structure commonly associated with each product, the documentation a payer expects, packaging and inventory handling, and how reliably a patient can complete follow-up visits. Why this matters is simple: the option that fits your scheduling, authorization, and traceability process is often easier to run consistently than one chosen only on name recognition.
Key Takeaways
- Both products are in the same class, but they are not the same product.
- Series design often drives clinic choice more than headline claims of relief.
- Coverage decisions are product-specific and payer-specific, including Medicare pathways.
- Documentation should tie diagnosis, prior conservative care, laterality, and injection history together.
- Safety monitoring is similar across the class and should include reassessment triggers after injection.
This briefing is written for licensed clinics and healthcare professionals.
Hyalgan vs Euflexxa: What Actually Differs
No, Euflexxa is not the same as Hyalgan. They belong to the same broad therapeutic category, but they are distinct products with different labeling, supply configurations, and common series expectations. That matters operationally. A prior authorization submitted for one brand does not automatically cover the other, and a clinic should not assume that mid-series substitution is acceptable unless the payer and treating clinician support it.
If you are reviewing the specific product listings for Euflexxa and Hyalgan, verify the exact catalog details, packaging description, and current instructions for use before scheduling. Clinics should treat the product record, not the class name alone, as the unit of documentation.
| Comparison factor | Hyalgan | Euflexxa | Why clinics care |
|---|---|---|---|
| Product type | Hyaluronic acid viscosupplement | Hyaluronic acid viscosupplement | Same class does not mean the same product record or coverage pathway. |
| Typical series pattern | Often associated with a longer multi-visit course, subject to current label and payer rules | Commonly handled as a shorter multi-visit course | Visit count affects staffing, reminders, and completion risk. |
| Packaging workflow | Verify exact syringe configuration on the item record | Verify exact syringe configuration on the item record | Receiving, lot capture, and chart build should match the exact SKU. |
| Administrative handling | Do not assume interchangeability with other viscosupplements | Do not assume interchangeability with other viscosupplements | Brand-specific documentation reduces substitution errors and claim issues. |
| Evidence interpretation | Read alongside class and regimen data | Read alongside class and regimen data | Many publications discuss class-level outcomes rather than a definitive brand winner. |
One reason the comparison can sound more certain than it really is is that published evidence often evaluates hyaluronic acid products as a class or compares dosing patterns rather than settling a clean brand-by-brand superiority question. A practical review therefore asks a different question: which option can your clinic deliver consistently, document cleanly, and reassess on schedule?
Why it matters: The hardest part may be completing the intended series, not naming the brand.
How Regimen Design Changes Program Fit
In day-to-day operations, Euflexxa and Hyalgan differences are often most visible at the front desk. A shorter standard series can reduce missed-visit exposure, simplify injection-room planning, and compress follow-up into a tighter time window. A longer series may still work well in practices with stable return rates, strong reminder systems, and dependable room turnover.
When fewer visits may help
Fewer planned encounters can matter when your population has transportation barriers, variable attendance, or heavy rescheduling pressure. It can also reduce the number of authorization-sensitive touchpoints and the amount of staff time spent coordinating bilateral treatment sequences. That does not prove better symptom control. It simply means the pathway may be easier to complete from start to finish.
Completion matters because an incomplete viscosupplementation course may blur both outcome tracking and billing follow-through. If your clinic measures response at the series level, every missed visit complicates the record. A product with a simpler visit cadence may therefore fit a busy knee OA service even when the clinical class is otherwise familiar.
When an established multi-visit workflow still works well
If your clinic already runs repeat injection pathways smoothly, a longer course may be operationally acceptable. Some practices prefer the cadence because it creates structured reassessment points, predictable note intervals, and room to pause if a payer asks for more records. The right fit depends on your process maturity, not only on the product label.
For a broader category view, the Joint Injections hub shows how knee OA programs may review related options alongside Monovisc, Synvisc One, Durolane, Supartz, and Orthovisc. That wider comparison is useful when the real question is single-visit versus multi-visit program design rather than one brand against another.
Patient Selection, Safety, and Reassessment
There is no universal best knee injection for osteoarthritis. The better question is which option fits the documented diagnosis, prior conservative management, symptom pattern, and the clinic’s ability to complete the intended course. Many payer policies ask for evidence that noninvasive treatment has already been tried and that the requested product aligns with labeled use. Your internal pathway should reflect that before the first syringe is prepared.
At the clinical level, both products sit in the same therapeutic family, so class-level counseling is often similar. Common issues after a hyaluronic acid injection can include temporary knee pain, swelling, warmth, stiffness, or effusion (fluid buildup in the joint). Those effects are usually handled as post-procedure monitoring points, not as proof that one brand failed. Any rapidly worsening pain, fever, marked redness, or inability to bear weight after an injection warrants prompt evaluation for infection or another complication.
It also helps to separate expected post-injection flare from true escalation signs. Mild transient discomfort or fullness in the joint may be documented and observed under clinic protocol. A severe change in symptoms, systemic illness, or pronounced local inflammatory findings should move the case out of routine follow-up and into a higher-acuity review pathway.
- Diagnosis fit — documented knee OA and symptom burden.
- Conservative care history — prior therapy, medication, or bracing noted.
- Series completion — patient can return for planned visits.
- Prior response — earlier injections and tolerance are recorded.
- Contraindication review — confirm current label and clinician protocol.
When comparing the two brands, remember that noninterchangeability is both a clinical and an administrative issue. If a patient starts one product, changing to another partway through the sequence may create charting gaps, claim denials, or uncertainty in outcome tracking. Standardize the pathway first, then measure results against that pathway.
Coverage, Documentation, and Workflow
Medicare coverage for Hyalgan injections and other viscosupplements is not a single national yes-or-no rule for every clinic. In practice, payment often turns on local policy language, prior conservative therapy requirements, interval rules, and whether the requested brand matches the documentation submitted. Commercial plans can be just as product-specific. For that reason, the comparison also becomes a paperwork question: the more tightly your template matches the exact product and series, the lower the chance of avoidable resubmissions.
Most clinics benefit from separating three records: the clinical rationale, the authorization packet, and the administration log. That structure helps when a payer asks for diagnosis history while your receiving team needs lot and expiration data for traceability. It also makes repeat courses easier to audit later, especially when a clinic stocks more than one knee gel injection option.
Coverage checks should also account for plan type. Traditional Medicare, Medicare Advantage, and commercial policies may not use identical documentation language, even when the underlying clinical question looks similar. If a clinic changes from one viscosupplement brand to another, update the authorization packet rather than assuming prior approval language still applies.
Questions to settle before scheduling
- Diagnosis support — imaging and clinical findings are documented.
- Payer criteria — product name and interval rules are confirmed.
- Laterality plan — right, left, or bilateral treatment is clear.
- Conservative treatment record — prior measures are easy to retrieve.
- Product traceability — lot, expiration, and receipt records are ready.
- Administration note — route, site, and follow-up plan are standardized.
- Repeat-course history — prior dates and response are visible.
Quick tip: Match the prior-authorization request to the exact brand and planned series language.
Brand-name product examples can be sourced through vetted distributor networks.
Storage and handling should follow the current instructions for use for the exact item you stock. Policies vary by clinic, but receiving teams typically document intact packaging, expiration date, and lot information at receipt and again at administration. If your program keeps multiple viscosupplement brands on hand, separate shelf labels and barcode workflows can reduce selection errors on injection days.
Where Other Viscosupplements Fit in a Knee OA Program
If your committee is reviewing the full knee OA pathway, not just two brands, keep the comparison framework consistent. Ask which products match your visit capacity, authorization resources, receiving process, and clinician preference for follow-up cadence. That is usually more useful than asking which name is best in the abstract.
Additional options in the category include Synvisc Classic, Cingal, and Hymovis. Clinics should review each product on its own label, packaging, handling instructions, and payer history. The fact that multiple products belong to the hyaluronic acid class does not make them administratively interchangeable.
- Visit burden — how many scheduled encounters your team can support.
- Authorization friction — whether payers recognize the product readily.
- Inventory format — how the item is packaged and documented.
- Outcome tracking — whether your registry compares like with like.
Verified supply channels support routine traceability and receiving checks.
Do not mix class comparison with unrelated injection categories. Corticosteroid injections, biologic products, and surgical pathways answer different clinical questions and should be compared on separate protocols. When clinics blur those categories, the discussion shifts away from workflow fit and into apples-to-oranges decision making.
Putting the Comparison Into a Clinic Decision Framework
For most practices, this choice is less about declaring a winner than standardizing a repeatable program. Choose the option that aligns with your typical patient return rate, authorization staffing, stocking model, and method for documenting repeat courses. Then audit completion rates, claim rework, and post-injection follow-up rather than relying on informal impressions.
A simple rule helps. If your main constraint is appointment burden, a shorter common series may fit better. If your main strength is an established repeat-visit pathway with dependable follow-up, a longer series may still integrate well. Either way, confirm current labeling, payer criteria, and clinic policy before scheduling. That is usually the clearest route to a stable knee OA injection program.
Authoritative Sources
- AAOS background on viscosupplementation appears in this AAOS overview of knee arthritis injections.
- Coverage checks should begin with the CMS Medicare Coverage Database.
- Evidence on regimen patterns is reviewed in this NIH-indexed comparative dosing analysis.
Both products can fit knee OA care, but the safest comparison is the one tied to label, documentation, and clinic workflow rather than brand shorthand alone.
This content is for informational purposes only and is not a substitute for professional medical advice.