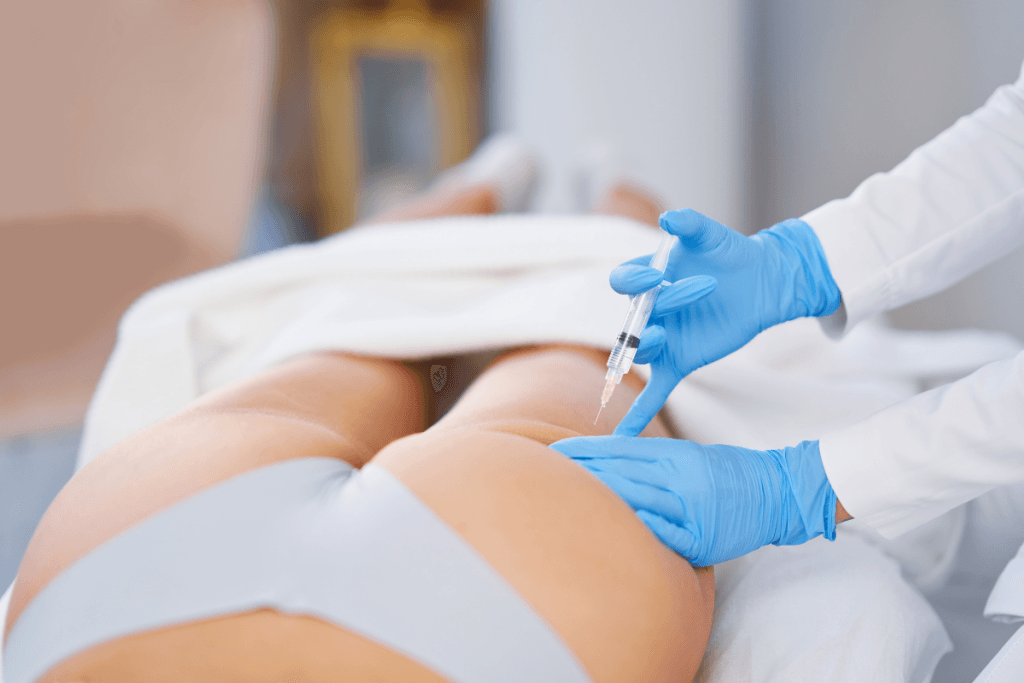
A body contouring supplies checklist helps clinics separate fixed equipment, treatment-specific accessories, consumables, infection-control items, and documentation tools before stock problems affect the schedule. The aim is not to carry every device, product, or accessory. The aim is to match each item to a live protocol, a trained provider, a storage requirement, and a clear reorder point.
This briefing is written for licensed clinics, practice managers, and procurement teams adding or standardizing body contouring services. It focuses on practical stock control: what belongs in the room, what should stay in backup inventory, how to document use, and how to judge whether a supplier fits clinical workflow. MedWholesaleSupplies serves licensed clinics and healthcare professionals, so the emphasis here is professional procurement rather than consumer treatment preparation.
Key Takeaways
- Define modality scope before purchasing devices, accessories, or products.
- Separate room infrastructure, consumables, sanitation items, and product-specific stock.
- Build room kits around active protocols, not aspirational service menus.
- Track lot, expiry, storage, cleaning, and maintenance in one routine.
- Use supplier verification and par levels to protect service continuity.
Build the Checklist Around Real Clinic Workflow
A body contouring supplies checklist should start with workflow, not with a catalogue. Body contouring can include noninvasive devices, energy-based treatments, injectable or biostimulatory protocols, and adjacent skin or cellulite services. Each pathway changes room setup, infection-control needs, documentation, and restocking cadence.
Start by naming the services your team can actually deliver today. Then map each service from consultation through room reset. Ask what must be present before the appointment starts, what gets used during treatment, what must be captured in the record, and what must be cleaned, quarantined, discarded, or reordered afterward. This process usually creates a more reliable body contouring supply list than copying a generic starter kit.
For broader service planning, the Body Contouring category can help teams review related educational topics. Product browsing should stay separate from protocol design, but the Body Contouring Products collection may help procurement teams understand how varied the category can become.
Start With Modality Scope
Each modality creates different supply demands. A radiofrequency or ultrasound workflow may rely on device accessories, conductive media, patient positioning items, approved cleaning products, and service logs. An injection-based workflow adds sharps handling, aseptic setup, lot capture, expiry review, and tighter separation of product-specific stock.
Why it matters: Standardized room setups reduce omissions and make staff coverage easier.
Clinics should also separate core body sculpting supplies for clinics from optional items used only in marketing demonstrations, photography, or occasional consultations. If a product or accessory does not connect to a current protocol, staff competency, or safety step, it usually does not belong in opening inventory.
Equipment and Room Setup Essentials
The equipment needed for body contouring is best viewed in layers. Clinics need a consistent treatment room foundation, modality-specific equipment, storage and tracking tools, and safety items that support turnover between patients. This approach keeps purchasing practical and makes audits easier.
| Category | What to Include | Stock Control Purpose |
|---|---|---|
| Core treatment room | Treatment bed, stool, task lighting, carts, measuring tools, and photography setup if used | Creates a repeatable room standard across providers and shifts |
| Modality-specific equipment | Device console, handpieces, applicators, approved tips, cables, pads, or accessories tied to the protocol | Links the room setup to the service instructions and maintenance plan |
| Injection support items | Skin marker, syringes, needles or cannulas if applicable, gauze, antiseptic, and sharps disposal | Supports only workflows that include injection-based steps |
| Product storage | Dedicated shelving, locked storage when appropriate, temperature checks when required, and expiry logs | Reduces mix-ups and improves traceability |
| Infection prevention | Hand hygiene supplies, gloves, barriers, surface disinfectant, waste bins, and linen handling items | Keeps rooms ready for safe turnover |
| Maintenance support | Approved cleaning materials, spare accessories, service records, and downtime notes | Prevents missing small parts from stopping the schedule |
A body contouring equipment checklist should distinguish capital purchases from recurring supplies. Clinics often choose a device before they fully estimate the accessories, barriers, cleaning agents, and documentation materials needed to use that room every day. Reliable stock depends on repeatability, not only on the launch purchase.
Use a room-by-room layout if more than one provider shares stock. Label carts consistently, keep treatment-specific items together, and make backup stock visible without allowing rooms to draw from each other informally. Informal borrowing is one of the fastest ways to lose count accuracy.
Consumables, Disposables, and Sanitation Stock
The body contouring consumables list is where daily reliability usually succeeds or fails. Clinics rarely cancel because a major device disappears. They lose time because small items run out, the correct accessory was opened in another room, or the cleaning shelf was not reset after a busy session block.
Build consumables around room kits. A room kit should contain the routine items needed for one workflow, with a separate central backup shelf for replenishment. This avoids mixing clean stock, opened items, near-expiry items, and procedure-specific products in the same drawer.
Consumables to Standardize
- Skin prep items — antiseptic, gauze, applicators, dressings, and marking tools when needed.
- PPE and barriers — gloves, masks or eye protection per protocol, drapes, and protective covers.
- Device-use extras — conductive gel, coupling media, replacement pads, tips, or approved accessories.
- Measurement tools — tapes, photo markers, and cleaned reusable tools when progress tracking is documented.
- Waste handling — sharps containers, clinical waste bags, spill supplies, and segregation labels.
- Cleaning stock — surface disinfectants, wipes, hand hygiene products, and linen handling supplies.
For injection-based pathways, maintain aseptic technique, meaning a clean method intended to reduce contamination. Keep sharps disposal visible and physically separate from clean stock. Product-specific procedure steps should live in the treatment protocol, not in a generic body contouring supplies checklist.
Clinics that offer injection-led contouring services may also need adjacent procurement planning. For example, Fat Dissolving Injections provides broader context for how this category differs from device-only pathways. If cannulas or needles are part of a protocol, review procurement and handling considerations through Cannulas and Needles Wholesale.
Quick tip: Separate opening stock from safety stock on every room count sheet.
Set par levels for high-turn items and rotate by expiry, not by shelf position. Par levels are the minimum and maximum quantities a clinic keeps on hand for routine service continuity. Review them after real usage data appears, because first-month estimates often miss the true rate of gloves, wipes, dressings, and single-use accessories.
Documentation, Consent, and Inventory Controls
A body contouring supplies checklist is incomplete if it only names products. Clinics also need records showing which item was used, where it was used, who used it, and whether it matched the consented service. The exact record format will vary by jurisdiction, service type, and internal policy.
At minimum, maintain a master SKU list, approved protocol list, consent forms matched to each service, lot and expiry capture where relevant, incident documentation, and cleaning or maintenance logs. If photography or circumference measurement is part of the clinic workflow, document permissions and storage rules separately from general consent.
- Define live services and limit opening stock to protocols staff can already support.
- Verify supplier and SKU by checking product identity, packaging, lot visibility, and instructions.
- Receive and inspect stock for seal integrity, quantities, and handling notes.
- Store by workflow so room kits, backup stock, and near-expiry items stay separate.
- Document use by capturing lot, room, provider, and accessory details when required.
- Audit and reorder through scheduled review of par levels, incidents, returns, and expiring items.
This structure matters when a clinic crosses between fat-dissolving, cellulite-focused, and body-volume categories. Some lines may be stored, documented, and consented differently. Product pages such as Aqualyx 10 8 mL Vials or HYACORP Body Contouring MLF 2 should be treated as SKU-specific references, not as substitutes for local protocols or professional judgment.
Keep naming consistent across the shelf label, inventory system, consent form, and treatment record. Marketing shorthand may help internal conversations, but procurement and charting need controlled names. This reduces mismatch errors and makes stock audits faster.
Supplier Review and Restocking Discipline
Reliable stock comes from supplier fit as much as product selection. A clinic needs sources that support licensed procurement, traceability, and repeat ordering. A large catalogue alone does not solve documentation gaps, unclear packaging, or inconsistent item naming.
Before adding any new SKU, confirm that the item fits an active service, staff have reviewed relevant instructions, storage conditions are clear, packaging supports lot capture, and the supplier can explain source verification. MedWholesaleSupplies works with vetted distributor networks and verified supply channels for licensed clinics, which is relevant when procurement teams need traceable professional sourcing.
A supplier review should also cover discrepancy handling. Ask how damaged packs should be quarantined, which records the clinic should retain, and what staff should do if packaging, labeling, or quantities do not match expectations. Policies vary, so keep these steps inside your clinic’s standard operating procedures.
For procurement workflow beyond body contouring, the Wholesale Procurement category can support broader stock-planning decisions. Teams building a wider aesthetic room standard may also find the Esthetician Supplies Checklist useful for comparing shared room consumables.
Planning Startup Stock for a New Service Line
Clinics starting a body contouring service usually benefit from a narrower opening plan. One documented modality with a clear expansion path is easier to train, audit, and restock than a crowded starter order.
Define room turnover time, provider training, cleaning steps, photography or measurement workflow, consent forms, and reorder ownership before adding more SKUs. Revenue expectations are not a procurement plan. Operational consistency is the safer starting point.
- One live modality — prove room flow before expansion.
- One room standard — use the same cart, labels, and count method.
- One reorder owner — assign routine stock review responsibility.
- One incident pathway — quarantine questionable stock and document the event.
- One review calendar — update consents, logs, and near-expiry checks on schedule.
Teams comparing noninvasive, injectable, and combination pathways can review Body Contouring Treatments for service-category context. Final supply decisions should still reflect local scope, device instructions, clinician training, and applicable professional standards.
Authoritative Sources
- For injection safety principles, see the CDC injection safety overview.
- For bloodborne pathogen requirements, review the OSHA Bloodborne Pathogens Standard.
- For device safety reporting context, review the FDA medical device reporting information.
Reliable clinic stock is less about owning more items and more about matching each item to a live workflow. A strong body contouring supplies checklist defines scope, standardizes room kits, documents product use, and keeps supplier review visible as volume changes.
This content is for informational purposes only and is not a substitute for professional medical advice.