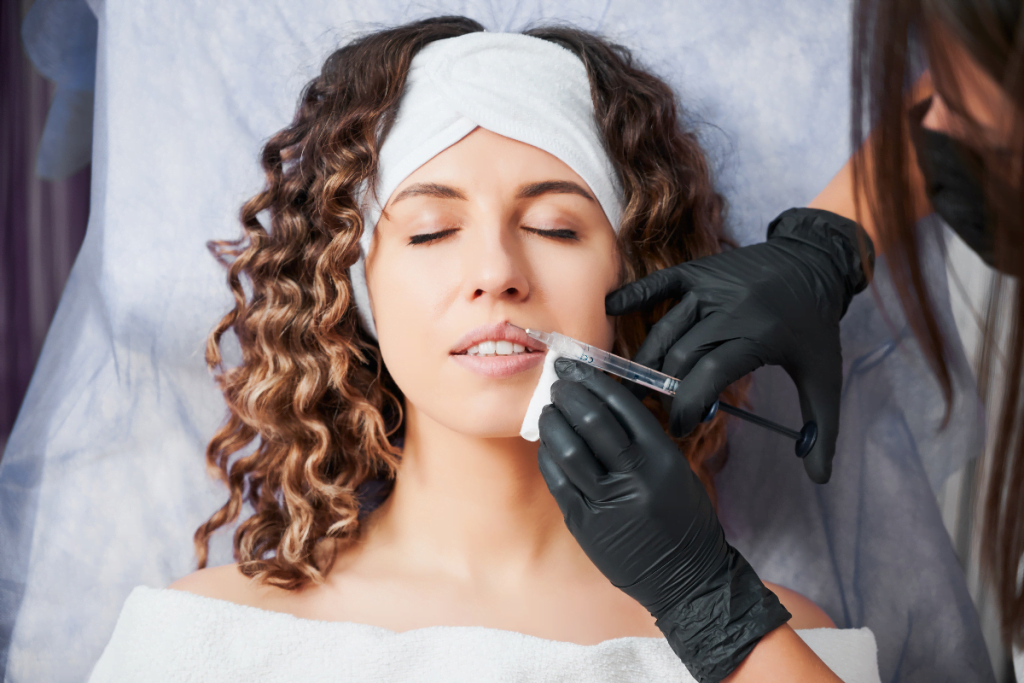
Can Radiesse be used on lips? For direct implantation into the lip body, the practical clinic answer is generally no. RADIESSE is a calcium hydroxylapatite filler, and official safety information states it should not be implanted in the lips. The distinction matters because many patient requests for “lip filler” are actually requests for perioral support, border refinement, or lower-face rejuvenation around the mouth.
This clinic briefing separates direct lip augmentation from adjacent perioral planning. It explains how the material behaves, where confusion starts, and how licensed teams can document evaluation, consent, follow-up, and alternative product selection without turning a vague cosmetic request into an unclear treatment plan.
Key Takeaways
- Direct lip placement is not the usual role of RADIESSE.
- Perioral support is different from filling the vermilion lip.
- Material properties and reversibility should guide product choice.
- Consent should name the exact anatomic target and labeling status.
- Follow-up plans should include expected effects and urgent warning signs.
This resource is written for licensed clinics and healthcare professionals.
Why Direct Lip Placement Is Usually Not Appropriate
Direct lip augmentation requires a product that can move softly with thin, highly mobile tissue. The vermilion lip is visible, tactile, and active during speech, eating, smiling, and compression. Small contour irregularities can be obvious to patients and clinicians because the area is central and frequently in motion.
RADIESSE is a calcium hydroxylapatite, or CaHA, filler. It is often discussed for structural support and collagen stimulation in selected facial areas. Those properties do not automatically fit soft lip volume. A firmer structural implant can feel less forgiving in the lip body than a softer hyaluronic acid filler, especially when the goal is pliable fullness or subtle border blending.
The more precise question is not simply whether RADIESSE can create a fuller mouth. It is whether the planned tissue target is the lip itself or the surrounding support frame. A patient may say “lips” while pointing to oral commissures, marionette-region support, or vertical perioral lines. That language gap can affect product choice, consent, photography, procedure notes, and aftercare instructions.
Why it matters: A lip request must be translated into anatomy before any filler is selected.
For background on CaHA behavior, the internal resource on Calcium Hydroxylapatite Filler reviews the class in broader aesthetic planning. For lip-specific technique concepts, see Lip Augmentation Techniques.
How Calcium Hydroxylapatite Behaves Around the Mouth
CaHA fillers provide a scaffold effect in tissue and may support collagen formation over time. This is why clinicians often discuss them in the context of structure, contour, and biostimulation rather than pure hydration or soft mucosal volume. Around the mouth, that distinction becomes clinically important.
The perioral region is not one uniform target. The lip body, vermilion border, cutaneous upper lip, oral commissures, chin-labial area, and lower-face support structures all behave differently. A product that suits deeper support in one zone may not suit superficial, mobile, thin tissue in another. That is why the phrase Radiesse lips can be misleading unless the chart specifies the exact treatment plane and objective.
Collagen stimulation also has a different clinical purpose than immediate lip plumping. When patients request a plush or hydrated lip, a biostimulatory filler may not match the desired tactile outcome. When they request improvement in support around the mouth, the clinician still needs to determine whether the target is a labeled area, an off-label consideration, or better handled with another modality.
Soft volume versus structural support
Soft volume usually means the patient wants a compressible, natural-feeling change inside the lip or along the border. Structural support means the clinic is evaluating the framework around the mouth, such as commissure support or adjacent fold softening. These are different treatment questions. They should not share the same shorthand in documentation.
Clinics comparing biostimulatory products can review Collagen-Stimulating Fillers for broader context. For product-family comparison, Radiesse and Juvederm Voluma covers structural filler decision points outside the lip-specific question.
Evaluation Before a Radiesse Lip Request Enters the Plan
A safe evaluation starts by defining the patient’s goal in tissue terms. The clinic should identify whether the request is for volume, border definition, vertical line softening, hydration, commissure support, or broader lower-face balance. Only then should product class and labeling status enter the treatment discussion.
If the request is direct lip enlargement, many clinics will consider a hyaluronic acid pathway instead. HA fillers are commonly used for lip volume because they can be selected for softness, shaping behavior, and adjustability. That does not mean every HA filler suits every lip. It means the material class often aligns more naturally with the tissue goal than a CaHA product.
History-taking should include prior filler placement, unknown or permanent filler history, previous inflammatory reactions, prior vascular events, oral herpes history when relevant, dental procedures, smoking status, and active infection or inflammation near the treatment zone. Planned dental work or unresolved periodontal concerns can complicate swelling, discomfort, and interpretation after treatment.
The physical exam should include tissue thickness, lip volume at rest, animation, smile pattern, commissure position, perioral line depth, chin-labial support, dental or prosthetic changes when relevant, and baseline asymmetry. A patient seeking larger lips may actually need a lower-face assessment before any lip-focused treatment is offered.
Contraindications and precautions
Contraindication review should follow current product labeling, local regulations, professional training, and clinic policy. Active lesions, mucosal irritation, uncontrolled skin inflammation, or infection near the intended area commonly prompt deferral in aesthetic workflows. If any contemplated use falls outside labeling, document that status clearly and explain the alternatives reviewed.
Risk discussion should include common injection-related effects such as tenderness, bruising, swelling, redness, asymmetry, and palpable change. It should also cover less common but serious concerns, including vascular compromise, infection, inflammatory reactions, and visual symptoms that require urgent escalation under a clinic complication protocol.
When procurement context is relevant, clinics should verify that products come through vetted distributors and verified supply channels. MedWholesaleSupplies serves licensed clinics and healthcare professionals in a B2B model, so product navigation should sit inside normal credentialing and documentation controls rather than patient-directed access language.
Consent and Documentation Workflow
Clear documentation protects both clinical reasoning and patient expectations. The note should state the exact anatomic target, the desired tissue effect, the product class considered, and why direct lip implantation was avoided if the patient asked for it. Avoid writing “lips” when the plan is actually perioral support.
For direct lip volume, the chart may state that a softer HA option was discussed because the goal involved pliability, adjustability, or vermilion shaping. For perioral support, the note should specify the treated area and whether the plan is on-label or off-label. Consent should not blur lip body augmentation with lower-face support.
A concise clinic workflow can standardize the conversation:
- Define the visible concern and anatomic target.
- Separate vermilion lip goals from perioral goals.
- Review labeling, contraindications, and precautions.
- Discuss alternatives and reversibility differences.
- Capture baseline photos and animation notes.
- Record product, lot, expiry, and injection site details.
- Provide written aftercare and escalation instructions.
Quick tip: Use separate chart phrases for lip volume and perioral support.
For broader browsing, the Dermal Fillers Category groups educational filler content. The Dermal Fillers Catalog can support product-family review for licensed clinic teams, but it should not replace label review or clinical judgment.
Alternatives for Volume, Definition, and Hydration Goals
The best alternative depends on the tissue objective. If the goal is soft lip volume, clinics often evaluate lip-appropriate hyaluronic acid fillers. If the goal is border refinement or fine etched lines, the plan may involve lower-volume precision work. If the goal is surface quality or hydration, a skin-quality approach may be more appropriate than structural filler.
Radiesse vs hyaluronic acid lip filler is therefore not only a brand comparison. It is a material-fit question. HA products can vary in firmness, cohesivity, spread, and intended use, while CaHA products are generally considered more structural and biostimulatory. The clinical decision should account for labeling, tissue thickness, desired feel, reversibility, prior filler history, and patient tolerance for staged correction.
Product examples can help teams frame the discussion without implying interchangeability. Restylane Kysse is a lip-associated HA product page. Juvederm Volbella is another HA product page often reviewed in lip and perioral planning contexts. Clinics should verify current labeling and suitability before any product selection.
For structural CaHA product reference, the Radiesse 1.5 mL page provides a product navigation point for licensed practices. Use that type of page as an inventory and specification reference, not as a substitute for the official instructions for use.
Recovery, Reassessment, and Escalation Planning
After filler procedures around the mouth, early swelling and tenderness can make immediate assessment unreliable. The region moves constantly, so contour changes may evolve as swelling settles and normal expression returns. Reassessment should compare baseline photos, rest and animation views, palpation findings, and the original tissue goal.
Questions about session count need careful framing. There is no universal number of syringes or sessions for a Radiesse lip request because the request may not be an appropriate lip treatment at all. If the actual target is perioral support, the number of sessions depends on baseline anatomy, product choice, tissue response, prior treatment history, and the clinic’s staged planning approach.
Patients should receive written instructions that distinguish expected post-injection effects from urgent warning signs. Severe pain, blanching, dusky or mottled skin color, rapidly worsening swelling, unusual visual symptoms, spreading redness, fever, or drainage should trigger prompt clinical review under the practice’s complication protocol. Staff should know who triages after-hours concerns and how escalation is documented.
Maintenance discussions should avoid promising a fixed timeline. Product behavior, tissue movement, metabolism, injection site, and treatment objective all influence follow-up planning. A clinic can explain that structural support, soft lip volume, and skin-quality goals may require different review intervals and different products.
Practical Bottom Line for Clinic Teams
The clearest answer to can Radiesse be used on lips is that direct lip implantation is generally not the right role for this CaHA filler. If the patient’s concern involves the perioral frame, evaluate the surrounding anatomy and document the target precisely. Do not let the word “lips” stand in for several different treatment goals.
A strong workflow moves in this order: define the anatomy, identify the tissue effect, review labeling, compare material behavior, discuss alternatives, document consent, and plan follow-up. That sequence helps clinicians avoid mismatched products, unclear notes, and unrealistic expectations.
For further product-class context, the internal article on Radiesse Collagen Support explains the biostimulatory discussion in broader terms. Keep the lip decision separate from the general collagen-stimulation discussion.
Authoritative Sources
- FDA RADIESSE patient information guide
- FDA RADIESSE device information
- American Society of Plastic Surgeons dermal fillers overview
Further review should combine current product labeling, a full lower-face assessment, and a complication plan that matches your clinic’s filler protocols.
This content is for informational purposes only and is not a substitute for professional medical advice.