Key Takeaways
- Product class: A viscosupplement (joint-lubricating injection), not a corticosteroid.
- Clinical use: Commonly discussed for symptomatic knee osteoarthritis care pathways.
- Expectation setting: Response and timing vary; document realistic goals and follow-up plans.
- Safety lens: Differentiate expected post-injection flare from red-flag presentations.
- Operations: Use verified sourcing, lot tracking, and label-based storage controls.
Overview
In orthopedic and sports medicine workflows, durolane hyaluronic acid is usually positioned as a viscosupplementation option for knee osteoarthritis (OA). This page summarizes what clinic teams tend to need day to day: what the product is, how it differs from steroid injections, what “gel injection” means in plain language, and how to anticipate post-procedure complaints without over- or under-escalating.
Why this matters: operational missteps often involve communication gaps, not technique. Clear documentation, standardized aftercare instructions, and consistent adverse-event triage reduce avoidable callbacks. For navigation to related inventory groupings, see Joint Injections as a clinic-facing browse hub.
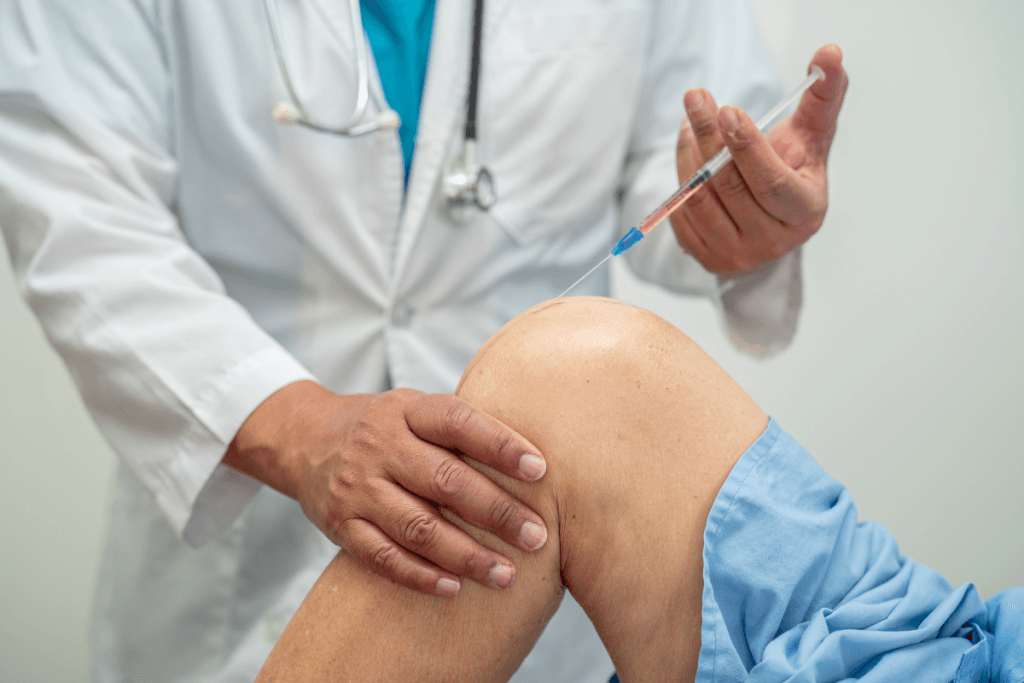
Durolane Hyaluronic Acid: Clinical Snapshot
This product is generally described as a single-session intra-articular (into the joint) hyaluronic acid formulation used for knee OA symptom management. In practice, clinics treat it as a device-like therapy with a defined labeling framework, rather than a compounded medication. Teams should align intake screening, consent language, and follow-up timing to the product’s official instructions for use and local standards.
Note: Many patient-facing sites describe these as “gel shots.” In clinical terms, that usually refers to viscosity and elastic properties of the injected material, not a different route or tissue target.
Core Concepts
What It Is (and What It Is Not)
Viscosupplementation aims to supplement synovial fluid properties in an osteoarthritic knee. The core ingredient is hyaluronic acid (a naturally occurring glycosaminoglycan), often explained to patients as a “joint lubricant” or “cushion.” durolane hyaluronic acid fits within this category, so it is distinct from intra-articular corticosteroids (anti-inflammatory drugs) and from biologics such as platelet-rich plasma.
Clinically, “is this a steroid?” is a frequent pre-procedure question. A useful operational answer is simple and consistent: it is not a steroid injection, and it does not follow steroid contraindication logic. That said, it still has its own labeled risks, post-injection expectations, and documentation needs.
Composition and Manufacturing Language Patients Will Hear
Patients may ask what the product is made from and whether it is animal-derived. Staff should be prepared for common phrasing like “rooster comb” and “avian allergy,” since some other viscosupplements are associated with avian sources. Where possible, anchor the conversation to the labeling and the manufacturer’s description of the material and stabilization method. Avoid speculating about cross-link chemistry or contaminants beyond what is documented.
When clinics carry multiple injectable hyaluronan products across service lines, confusion increases. Dermal hyaluronic acid fillers and skin “hydration boosters” are formulated and regulated differently than intra-articular products. If your practice stocks dermatology injectables, you can highlight the separation by pointing staff to examples such as Hyaluronic Acid 35 for skin applications as a contrast, while keeping joint injections governed by their own IFU and credentialing pathways.
Mechanism Framing: Symptom Modulation vs Structural Claims
Clinics often need a careful way to describe expected benefit without implying cartilage regeneration. Most authoritative materials frame viscosupplementation around symptom relief and functional improvement for some patients. Avoid statements that it “rebuilds cartilage” unless an official label explicitly says so. In chart notes, it helps to separate symptomatic goals (pain with stairs, walking tolerance, night pain) from structural disease language.
Plain-language summaries can be accurate and still conservative. “This is intended to improve joint fluid mechanics and may reduce symptoms for some people” is typically safer than absolute phrases. Documentation should also reflect what was tried before, and why this step was selected now.
Adverse Events and the “Pain Worse After Injection” Call
Teams should anticipate a predictable set of post-injection contacts: transient soreness, swelling, stiffness, and activity-related discomfort. Patients may describe this as “severe pain after the injection,” even when findings are benign. Intake scripts should capture timing, functional change, fever or systemic symptoms, and whether the joint looks progressively inflamed.
From a clinic-ops standpoint, the goal is consistent triage. Build a pathway that distinguishes expected post-procedure flare from presentations that warrant urgent assessment. Avoid giving blanket reassurance by phone. Instead, document symptoms, provide the clinic’s standard escalation options, and follow label-aligned precautions.
Setting Expectations on Onset and Durability
“How long does it take to work?” is common, and “durolane injection reviews” online can create unrealistic timelines. The safest approach is to acknowledge variability. Some patients report change quickly, while others notice gradual improvement over a longer interval. Your after-visit summary should avoid hard promises and instead specify what to monitor and when follow-up occurs in your practice model.
It also helps to clarify what “works” means. Patients may expect complete pain elimination. Consider documenting measurable endpoints such as reduced rescue analgesic use, improved function on a short scale, or improved tolerance for prescribed rehabilitation activities.
Practical Guidance
Operationalizing viscosupplement visits requires more than ordering the syringe. Standardize pre-visit screening, consent, procedure documentation, and post-visit instructions. If your clinic already uses structured documentation for other injectables, you can borrow that discipline. For example, the consent and photography workflows described in Sculptra Clinical Guide can inspire similar checklists, even though the indication differs.
For knee visits specifically, many clinics build a standing template that includes laterality, approach, ultrasound use if applicable, lot number, expiration date, and any immediate reaction. Include a brief note for staff on what patients may ask afterward, such as “can I drive after the injection?” or “can I walk after?” Keep answers aligned to your local policy and the product labeling rather than generic internet timelines. In that workflow, durolane hyaluronic acid should appear as the exact product used, with standardized nomenclature for billing and internal QA.
Clinic-Ready Checklist: Day-of Procedure Workflow
Use a short checklist to reduce variance between providers and locations. The goal is reproducibility, not rigidity.
- Confirm patient identity, laterality, and indication documentation.
- Review allergies and prior injection reactions in the chart.
- Verify product integrity, lot, and expiration before opening.
- Document aseptic prep steps per clinic policy.
- Record technique details used by the provider (site, guidance method).
- Provide written post-visit instructions and escalation criteria.
Handling Common Post-Visit Questions Without Overpromising
Patients commonly ask whether the injection is a “gel,” whether it contains steroid, and what activity limits apply. Staff should use consistent phrasing and avoid clinical debates at the front desk. If your practice also performs skin injectable procedures, it can help staff understand “same ingredient, different product class” by reviewing a comparator article like Jalupro Vs Profhilo, then explicitly noting that joint products follow separate labeling and training requirements.
Tip: Create a one-page phone script for “increased pain” and “swelling” calls. Include prompts for red flags, and a clear handoff to clinical staff.
Compare & Related Topics
Clinics often evaluate viscosupplements as a set, not as isolated products. Comparisons like durolane vs synvisc, durolane vs euflexxa, and durolane vs monovisc come up during formulary review. Patients may also ask questions framed as “which is better monovisc vs orthovisc.” Avoid simple winner/loser language. Instead, compare attributes that affect workflow, tolerability counseling, and payer rules.
In cross-service practices, patients may also confuse joint therapy with cosmetic injectables. It can help to differentiate hyaluronic acid joint therapy from biostimulants. For broader context on injectables that stimulate collagen (a structural protein), see Sculptra Vs Filler as a background read, then document in your knee OA notes that the mechanism and target tissue are different.
| Comparison area | What clinics document | Why it affects operations |
|---|---|---|
| Product type | Viscosupplement vs corticosteroid vs other | Sets expectation language and contraindication screening. |
| Regulatory labeling | Indication, warnings, IFU specifics | Determines consent elements and standardized aftercare sheets. |
| Administration pattern | Single-session vs series products (label-based) | Changes scheduling load and follow-up touchpoints. |
| Source and formulation | Cross-linking, avian vs non-avian (per label) | May influence allergy history prompts and patient education. |
Clinic Ordering and Compliance Notes
Procurement teams should align ordering, receiving, and inventory controls with the same rigor used for other implantable or injectable products. Use label-based storage requirements, consistent lot tracking, and documented chain-of-custody steps between receiving and clinical areas. For internal navigation, Orthopedic Injections can support formulary organization alongside your clinic’s policy library.
Ordering through MedWholesaleSupplies is restricted to licensed clinics and credentialed healthcare professionals. The business model is clinic-facing, with brand-name products supplied through screened distribution partners to reduce avoidable sourcing risk. For brand-level context used by some buyers during reconciliation, see Durolane Brand as a reference point for catalog alignment.
Also separate joint products from aesthetic stock to reduce selection errors. If your practice carries skin hydration products, store them in distinct bins and label clearly. A simple example is keeping dermal HA items like Viscoderm Hydrobooster outside the orthopedic injection workflow, since the intended tissue plane and clinical documentation differ.
Authoritative Sources
For claims, indications, contraindications, and adverse reactions, prioritize the official labeling and regulator summaries over blogs and forum posts. durolane hyaluronic acid should be referenced using the same name that appears on the IFU and any local policy documents to avoid ambiguity in audits.
- FDA PMA database for regulator-hosted device approval records and associated documents.
- AAOS OrthoInfo for high-level explanations that can help align patient education handouts with orthopedic terminology.
Recap: This therapy sits within viscosupplementation, not steroid care. Strong clinic workflows focus on labeling, documentation consistency, and predictable triage of post-injection symptoms.
This content is for informational purposes only and is not a substitute for professional medical advice.
________________________________________________________________________________________
Medically Reviewed by: Ma Lalaine Cheng.,MD.,MPH