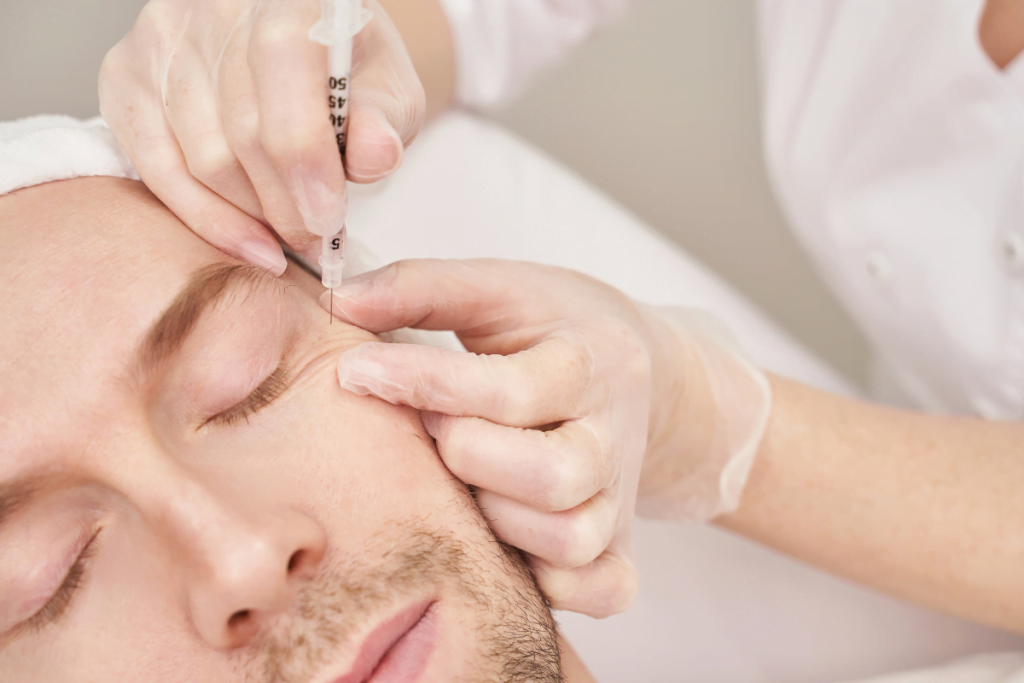
Yes, treatment can go wrong, although most post-injection changes are mild and self-limited. For clinics, the practical issue is triage: distinguish expected swelling, redness, bruising, and tenderness from red flags such as escalating pain, blanching, mottled discoloration, spreading warmth, discharge, or systemic allergic symptoms. This briefing on Profhilo side effects focuses on what is usually benign, what may suggest infection or vascular occlusion (blocked blood flow), and how to structure a documented clinic response.
This briefing is written for licensed clinics and healthcare teams.
Key Takeaways
- Most early reactions are local, mild, and trend downward rather than upward.
- Severe pain, color change, spreading erythema, discharge, or visual symptoms need same-day review.
- Document product, batch, sites, onset, photos, and escalation steps before symptoms evolve.
- Risk reduction starts with assessment, consent, aseptic technique, and clear aftercare instructions.
- Traceable sourcing and product-specific protocols support safer clinic response.
Profhilo Side Effects: What Is Usually Mild
Most Profhilo side effects are localized injection-site reactions. Mild swelling, small needle marks, limited redness, tenderness, and bruising can occur after any hyaluronic-acid procedure. In a routine course, these findings stay confined to the entry points, remain proportional to the treatment, and improve rather than spread. The useful question is not whether a mark exists, but whether the pattern fits a normal early tissue response.
Trajectory matters more than a single snapshot. Expected reactions usually peak early and then settle as the acute inflammatory response eases. Concerning reactions move the other way: pain increases, edema becomes tense, skin color changes, or a previously quiet area becomes warm and more indurated. That distinction should be part of staff training whenever a clinic handles Profhilo HL or body-focused options such as Profhilo Body Kit.
Short-lived surface irregularity can also look worse than it is, especially in thin skin or where edema peaks visibly. Appearance alone does not define a complication. What changes the response is progressive pain, evolving discoloration, marked asymmetry, or symptoms that return after an initially calm period. When clinics compare injectable HA categories, a broader Hyaluronic Acid Overview can help frame why tissue plane, hydration effect, and technique influence the early post-treatment picture.
Most mild reactions are concentrated in the first short post-treatment window and trend downward. If a symptom stays static for too long, worsens, spreads, or reappears after initial improvement, it deserves reassessment. That is often the clearest answer when teams ask how long ordinary post-treatment effects should last.
| Finding | Usual early pattern | What changes the response |
|---|---|---|
| Swelling and tenderness | Localized, early, and non-progressive | Escalating pain, tense edema, or new color change |
| Bruising | Visible at entry points with stable discomfort | Rapid expansion, marked pressure, or severe pain |
| Redness | Mild erythema near injection sites | Spreading warmth, increasing tenderness, or fever |
| Small lumps or papules | Limited surface irregularity that settles | Persistent nodules, warmth, delayed swelling, or pain |
Warning Signs That Need Same-Day Review
Same-day review is warranted when symptoms intensify, spread, or suggest tissue injury instead of ordinary post-treatment irritation. Severe pain, blanching, a net-like purple pattern, dusky color, rapidly increasing edema, drainage, or any new visual symptom should move the case out of routine aftercare and into active assessment.
Why it matters: Delay, not rarity, often drives the harm from injectable complications.
Possible vascular event
A vascular event is uncommon, but clinics should assume urgency when pain is disproportionate or skin perfusion changes. Sudden blanching, cool skin, slowed capillary refill, livedo, or progressive duskiness may indicate vascular compromise from intravascular placement or external vessel compression. Any visual disturbance is an emergency. If reduced blood supply persists, tissue ischemia can progress to ulceration or skin necrosis. In that sense, Profhilo can theoretically cause necrosis, even though the event is not a routine outcome.
Possible infection
Infection is more likely when redness spreads beyond the expected entry sites and becomes hotter, firmer, more tender, or fluctuant. Purulent drainage, malaise, or fever strengthen concern. Tiny needle marks alone are not infection. What matters is worsening inflammation, delayed onset after an initially calm period, or symptoms that feel increasingly unwell rather than cosmetically bothersome.
Hypersensitivity or delayed inflammation
Hypersensitivity can present with diffuse swelling, hives, wheeze, throat symptoms, or generalized flushing. Delayed inflammatory reactions may show up later as persistent edema, tenderness, or nodules after the first post-treatment window has passed. These cases still need prompt review because the management path differs from simple bruising or surface papules.
Front-desk triage scripts should reflect these differences. A staff member does not need to diagnose the event, but they do need to recognize when escalating pain, spreading redness, darkening skin, or systemic symptoms require clinician review rather than routine reassurance.
Clinic Response Protocol After an Unexpected Reaction
A useful clinic response starts by pausing further treatment and classifying the problem. Do not treat all unexpected reactions as if they are the same event. A stable bruise, suspected infection, allergic reaction, and possible vascular compromise each require different urgency, different documentation, and different escalation routes.
Policies vary by jurisdiction and supervision model, but the first operational steps are usually consistent. Clinics should have a written pathway, named contacts, and a clear threshold for urgent referral. Our broader Injection Safety hub can support staff training alongside local complication protocols.
- Stop and assess: Halt treatment, identify the treated area, and record when symptoms started.
- Check pattern: Note pain severity, color change, swelling progression, warmth, and any systemic symptoms.
- Assess urgency: Treat visual change, blanching, livedo, dusky skin, or severe pain as urgent.
- Document clearly: Record product name, batch, expiry, injection sites, volume, photos, and staff involved.
- Escalate promptly: Alert the prescriber or medical lead and follow the clinic complication pathway.
- Arrange review: Same-day in-clinic review, urgent specialist input, or emergency transfer may be needed.
- Close the loop: Record advice given, follow-up contact, outcome, and any required incident reporting.
Emergency stock planning may include adjunct items such as Hyaluronidase 1500 IU, but use should follow product-specific, prescriber-led protocols rather than a one-size-fits-all rule. The key operational point is readiness: the clinic should know who can review, who can authorize escalation, and where the documentation lives before a complication occurs.
Quick tip: Record photos and batch details before swelling or discoloration evolves.
Reducing Avoidable Risk Before Treatment
Reducing Profhilo side effects starts before the appointment. Risk screening should cover current skin infection, recent facial infection where relevant, prior reactions to injectables, medication and bruising history, immune or inflammatory conditions, recent procedures in the same area, and whether the planned site has thin tissue or difficult vascular anatomy. Baseline photographs and documented asymmetry help distinguish a new complication from a pre-existing contour issue.
Consent needs to be specific. Clinics should explain common injection-site reactions, the rare but serious possibility of vascular injury, infection, and allergic symptoms, and the exact route for after-hours escalation. Written aftercare should tell patients what is expected, what is not, and when the clinic wants a same-day update. Vague reassurance creates late presentations.
Technique and environment still matter. Aseptic prep, correct storage per manufacturer information, calm pacing, accurate site mapping, and avoidance of treatment through active dermatitis or broken skin all reduce preventable problems. Clinics should also separate who can advise remotely, who can perform urgent review, and who can authorize transfer or specialist referral.
Use vetted distributors and documented supply channels for injectable stock.
Traceability is part of safety, not just procurement. Batch number, expiry, opening details, injection points, photos, and the treating clinician should be easy to retrieve. That applies whether a clinic is working with Profhilo Structura, hydrating injectables such as Viscoderm Hydrobooster, or other HA-based products in the same service line.
Special Situations: Under-Eye Areas, Lumps, and Delayed Reactions
The under-eye area deserves a lower threshold for review. Thin skin, visible vessels, and a tendency to retain fluid make prolonged puffiness and contour irregularity more noticeable there than in broader facial areas. Not every under-eye lump is dangerous, but persistent edema, color change, marked tenderness, or new asymmetry should prompt reassessment rather than reassurance alone.
That matters when clinics compare periorbital options. Products such as PhilArt Eye, general skin-quality injectables such as Restylane Skinboosters Vital Light, or broader rejuvenation options such as Juvederm Volite do not share identical rheology, placement depth, or post-treatment feel. Staff should not assume that one area or one product class predicts every other response.
Lumps and nodules are also not all the same. Early superficial blebs may reflect temporary product visibility or local edema. Firmer, persistent nodules, delayed swelling after an initially quiet period, or tenderness with warmth deserve examination for inflammation, infection, or placement-related issues. Long-term problems are better defined by persistence and progression than by the mere fact that a patient can still feel the area.
Delayed problems also need classification. A late nodule may represent inflammatory change, biofilm-related infection, or an unrelated dermatologic issue. Reassess timing, intercurrent illness, recent procedures, and product traceability before labeling it a routine after-effect. This is where follow-up notes and photographs become clinically useful rather than administrative.
Brand-name injectable inventory should remain traceable in clinic records.
For clinics that stock multiple HA lines, the practical rule is consistency: keep product-specific instructions available, document anatomical plans carefully, and separate routine follow-up from emergency escalation. The same principle applies across facial and body protocols rather than relying on one generic aftercare script for every injectable service.
Authoritative Sources
- General aseptic practice is outlined by the CDC Injection Safety program.
- General soft-tissue filler risk warnings are summarized by the FDA dermal fillers safety page.
- Adverse event reporting routes are described by the MHRA Yellow Card scheme.
For clinics, Profhilo side effects are mainly a recognition and response issue. Most reactions are minor and settle, but severe pain, perfusion change, spreading inflammation, or systemic symptoms should trigger documented same-day escalation. Clear consent, traceability, staff training, and protocol-led follow-up do more to reduce harm than reassurance alone.
This content is for informational purposes only and is not a substitute for professional medical advice.