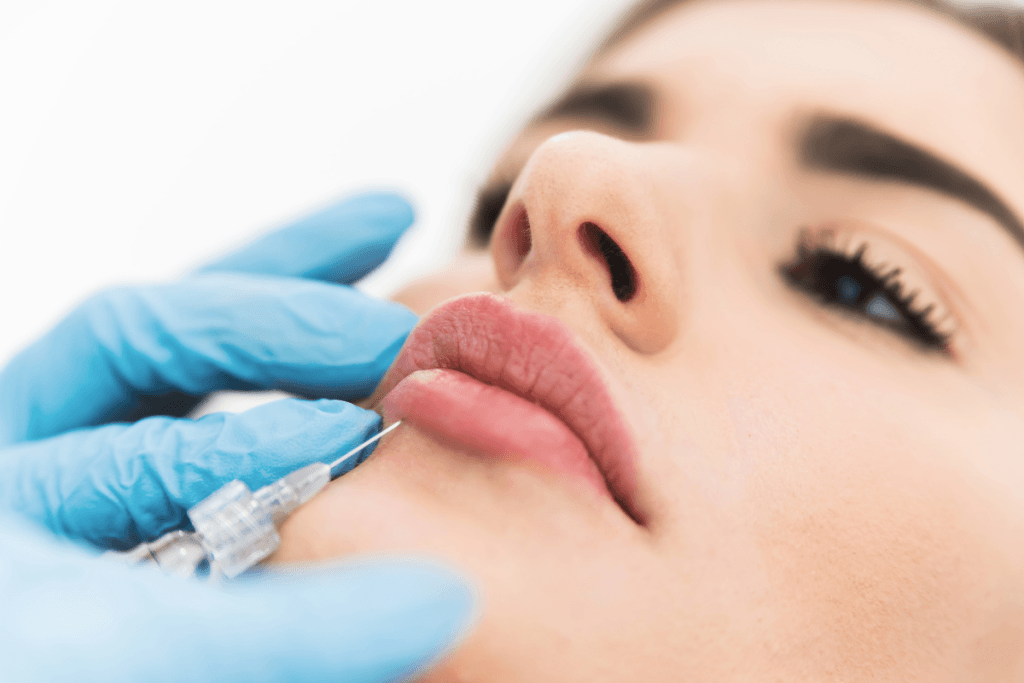
For clinics evaluating Aliaxin FL Lips for Clinics: Soft Volume and Shape Planning, the main issue is fit, not hype. A softer hyaluronic acid filler profile may suit subtle lip contouring, restrained volume, and selective treatment of perioral lines when anatomy, tissue quality, and safety readiness align. That matters because the lips are mobile, vascular tissue. Product choice influences contour control, swelling patterns, review timing, and complication management just as much as the treatment plan itself.
This discussion is framed for licensed clinics and healthcare teams.
Key Takeaways
- Start with the endpoint, not the brand.
- Assess shape, movement, and tissue quality first.
- Separate contour goals from bulk volume goals.
- Verify local regulatory status before sourcing or use.
- Document consent, photos, lot details, and escalation planning.
Soft Volume and Shape Planning for Clinics
Aliaxin FL is discussed within the Aliaxin hyaluronic acid filler range as a lip-focused option. In clinic planning, it is usually considered when the goal is softness, border refinement, and controlled shaping rather than a heavier volumizing effect. It may also enter the conversation when lines around the mouth need review alongside lip contour, because those concerns often appear together.
That does not make it the right choice for every lip case. Some patients want sharper definition at the vermilion border, some need more structural support, and some are poor candidates for treatment on that day because of inflammation, active infection, or unrealistic expectations. A good planning process starts with the endpoint: subtle enhancement, better symmetry, improved lip frame, or blended perioral detail. If the endpoint is unclear, product selection becomes guesswork.
A common planning error is expecting one filler choice to solve border definition, central body fullness, and etched perioral lines equally well. In many clinics, the better question is whether the case needs a staged plan, adjunctive review, or no treatment at all. Product fit is only one part of safe, coherent lip planning.
For wider category context, the Types Of Dermal Fillers resource and the browseable Dermal Fillers Hub help place lip-focused products in the broader facial filler landscape.
Assessment Points Before You Choose a Lip Filler
Before selecting any lip filler, decide whether the case is primarily about contour, volume, proportion, surface change, or correction of an earlier result. Those goals often overlap, but one usually dominates. Aliaxin FL lip filler planning tends to make the most sense when your priority is soft definition and measured shaping rather than maximal projection.
Lip anatomy and movement come first
Start with upper-to-lower lip balance, vermilion show, Cupid’s bow, philtral support, oral commissures, and smile dynamics. Static photos matter, but dynamic assessment matters more. A product that looks suitable on a still image may behave differently once the patient speaks, smiles, or purses the lips. Tissue thickness, prior filler history, scar burden, and baseline asymmetry all change how a softer filler may perform.
Perioral lines need separate thinking
Lines around the mouth can reflect volume loss, repetitive orbicularis oris activity, photodamage, smoking history, or changes in dental support. They should not be treated as a simple extension of lip volume planning. If surrounding support is weak, treating lines alone may leave the overall result unbalanced. Clinics should distinguish lip-body fullness from border refinement and from fine perioral creasing before any product is selected.
Candidate selection should stay practical. Review prior lip filler, episodes of prolonged swelling, cold sore history where relevant, bleeding risk, inflammatory skin issues, and recent procedures that could affect timing or tissue stability. This is also the point to identify patients who are chasing trends rather than realistic shape goals. When the request is vague, use reference photos carefully and document what the clinic is and is not aiming to change.
Why it matters: Small product-choice errors are more visible in lips than in many other areas.
For broader planning frameworks, see Facial Aesthetic Planning and Lip Augmentation Techniques.
Aliaxin LV and FL: Differences That Matter
When clinics compare FL with LV, the useful question is not which product is better. It is which product behavior best matches the planned endpoint. In broad product-line discussions, FL is often associated with softer contouring and fine-detail lip work, while LV is more often considered when fuller lip volume is the main objective. Exact labeling and availability can vary by market, so current product information should lead the decision.
| Decision factor | FL-style planning focus | LV-style planning focus |
|---|---|---|
| Primary aesthetic goal | Soft contour, balanced shape, restrained volume | More noticeable fullness or projection |
| Tissue behavior sought | Finesse in mobile lip tissue | Greater emphasis on volumizing effect |
| Perioral line overlap | May be considered when mouth-area detail also matters | Usually a secondary consideration |
| Assessment priority | Border control, symmetry, and movement | Central body fullness and structural support |
This comparison is only a starting point. In real practice, injector technique, tissue resistance, prior filler, and the patient’s tolerance for swelling can outweigh the product name. If your team is comparing filler classes rather than single brands, the Hyaluronic Vs Non-Hyaluronic Fillers overview is the better next read.
Safety, Contraindications, and Regulatory Questions
Safety planning matters as much as aesthetic planning. Lip tissue has dense vascular anatomy, frequent movement, and high visibility after treatment. That combination makes even minor errors obvious and makes serious complications time-sensitive. Clinics should screen for active local infection, uncontrolled inflammation, incompatible recent procedures, clinically relevant allergy history, and reasons to defer until the tissue is stable.
Teams also need a clear response plan for vascular occlusion, meaning blocked blood flow, as well as severe edema, delayed inflammatory reactions, and suspected herpes reactivation when clinically relevant. Emergency readiness is not just having supplies on site. It includes role clarity, documentation flow, escalation thresholds, and post-treatment instructions that tell the patient exactly what changes need urgent review.
Do not assume every Aliaxin product shares the same regulatory status in every country. Clinics should verify the current U.S. position, local labeling, and intended use before sourcing or treatment planning. Authorization in one market does not automatically translate to another. This matters especially when teams review international product literature or compare packaging across suppliers.
Counterfeit risk and packaging mismatch are operational issues, not just procurement issues. Clinics should verify lot identifiers, seal integrity, labeling language, and storage instructions against the product information supplied for their market. If anything is inconsistent, pause use until the chain of custody is clear.
Any sourcing discussion here assumes vetted distributor pathways.
Quick tip: Keep complication contacts, emergency supplies, and lot-trace documentation accessible in the same workflow.
For related operational reading, see Dermal Filler Safety Protocols, Hyaluronidase Workflow, and Sourcing Standards.
Clinic Workflow Snapshot for Soft Volume Planning
Operational consistency reduces avoidable variation. A simple workflow keeps lip planning centered on suitability rather than impulse selection.
- Verify product status and labeling for your market.
- Confirm goals, exclusions, prior filler history, and baseline photos.
- Document lip shape concerns, dynamic movement, and realistic endpoints.
- Prepare traceability records, consent language, and complication-readiness supplies.
- Plan review timing, aftercare counseling, and escalation instructions before treatment.
- Record the treatment rationale and follow-up findings in the same note set.
Documentation should explain why this filler category was selected for this case, what features were prioritized, and which trade-offs were discussed. In lips, those trade-offs often include softness versus structure, contour versus fullness, and immediate appearance versus early swelling. Baseline photography from multiple views helps the team judge outcome fairly and supports safer follow-up conversations.
Storage and handling should follow current label directions. Teams should document receipt, storage location, and any handling exceptions where applicable. Policies vary by supplier and jurisdiction, so internal procedures should align with current manufacturer information rather than habit.
For follow-up frameworks, review Post-Treatment Care Essentials.
What to Expect After Treatment and at Review
Most follow-up questions after lip filler treatment relate to swelling, bruising, early asymmetry, and whether the final shape matches the plan once movement returns to normal. Clinics should set expectations that the immediate appearance is not the final assessment point. Early review should distinguish expected short-term changes from warning signs that need faster escalation.
Review planning should separate urgent safety checks from later aesthetic reassessment. Early fullness may reflect edema rather than final shape. Aesthetic review should wait until the tissue settles enough to judge contour, symmetry, and movement reliably, based on clinic policy and current product guidance.
Aftercare counseling should be clear and brief. Advise patients how to contact the clinic, what symptoms warrant prompt review, and when a routine review is scheduled. Sudden blanching, escalating pain, dusky discoloration, marked temperature change, or visual symptoms need urgent clinical assessment. Those are not watch-and-wait findings.
Brand-name products should be checked through verified supply channels.
Where teams want broader context on durability counseling, further reading includes Lip Filler Duration. Final wear time varies by product, tissue behavior, technique, and patient factors, so counseling should stay product-specific and conservative.
Authoritative Sources
- FDA overview of dermal fillers and soft tissue fillers
- Official Aliaxin FL product information from IBSA Derma
- Peer-reviewed discussion of personalized facial filler protocols
In short, soft-volume lip planning works best when the filler choice follows the case, not the other way around. Further reading should focus on anatomy, documentation, complication readiness, and current labeling in your market.
This content is for informational purposes only and is not a substitute for professional medical advice.