Eylea HD in Retina Practice: Label, Safety, Workflow is best approached as a label-first operational topic for retina clinics. The central questions are indication fit, intravitreal-use safeguards, storage controls, and documentation after injection. That matters because the highest day-to-day risk often sits in scheduling, product integrity, sterile technique, and follow-up instructions rather than in a single ordering decision.
For most practices, the practical task is to translate the current FDA labeling into a repeatable clinic process. That means confirming approved use, screening for contraindications, separating routine ocular reactions from red-flag events, and making sure refrigeration, lot capture, and billing steps are not copied from another anti-VEGF product without review.
Key Takeaways
- Use the current FDA label as the source of truth for indication, contraindication, and scheduling details.
- Build workflow around vial integrity checks, aseptic preparation, lot capture, and documented storage conditions.
- Train staff to separate common ocular reactions from urgent events such as endophthalmitis or retinal detachment.
- Do not assume handling, room-temperature limits, or templates match other anti-VEGF products.
How Eylea HD Fits Retina Workflow
It fits as an intravitreal anti-VEGF therapy whose operational impact extends well beyond the injection room. Practices need a clear view of which diagnoses are on label, how the regimen is documented, and how staff distinguish product-specific instructions from class-wide habits.
At a high level, clinics usually review this formulation as aflibercept for intravitreal use under the current FDA label. As of current U.S. labeling, approved uses include neovascular wet age-related macular degeneration, diabetic macular edema, and diabetic retinopathy. For retina practices, that means referral intake, imaging review, consent language, and payer documentation should all point back to the exact labeled use being treated.
The important workflow point is simple. A retina clinic does not manage this product only at the moment of injection. It manages it across procurement, cold storage, scheduling, timeout, lot capture, adverse-event triage, and claim submission. Small template errors can become larger operational problems when multiple anti-VEGF products sit in the same refrigerator or share similar chart shortcuts.
| Topic | What the label answers | Why the clinic cares |
|---|---|---|
| Approved use | Which retinal conditions are on label | Supports correct intake, charting, and payer notes |
| Contraindications | When the product should not be used | Prevents injection-day delays and avoidable risk |
| Scheduling | How regimen timing is defined by indication | Keeps recall templates and follow-up intervals aligned |
| Storage | Refrigeration and excursion rules | Protects product integrity and inventory decisions |
| Administration | Intravitreal-use boundaries and precautions | Standardizes timeout, preparation, and documentation |
Label Points That Drive Daily Use
The label matters because it determines far more than eligibility. It shapes the clinic’s scheduling logic, consent workflow, product release from storage, and what staff should verify before a syringe ever reaches the field.
Indications and exclusions
The first check is whether the treated condition matches the current approved indication. In retina practice, that usually means confirming diagnosis coding, imaging support, and the treating physician’s documented rationale before product is assigned to a visit. A mismatch here creates downstream issues in scheduling, prior authorization, chart integrity, and billing.
Contraindications also belong upstream, not at the last minute. The current FDA labeling should be reviewed for exclusions such as ocular or periocular infection, active intraocular inflammation, and hypersensitivity concerns. Clinics should also avoid using product that appears expired, damaged, tampered with, or otherwise outside release criteria. That sounds basic, but packaging and storage failures are operational events, not just pharmacy events.
Scheduling and administration boundaries
Seen through Eylea HD in Retina Practice: Label, Safety, Workflow, the schedule question is not whether a single interval sounds familiar. It is whether the exact indication-specific regimen in the current prescribing information has been followed, documented, and communicated to staff. Clinics should not assume that a four-week pattern, loading concept, or follow-up cadence used elsewhere automatically applies here.
This is also why the common question about every-4-week use needs a careful answer. A retina clinic should look to the current package insert or prescribing information for the indication being treated, then align recall intervals and order templates to that source. The safer habit is to make the live label the scheduling reference of record, especially when a practice carries both newer and older anti-VEGF options.
MedWholesaleSupplies serves licensed clinics through B2B supply channels.
Safety Signals Clinics Need to Watch
The major safety burden is injection-related ocular risk plus the standard anti-VEGF caution set. Clinics should frame this in two layers: serious but less common events that need urgent recognition, and more common ocular reactions that still require clear counseling and documentation.
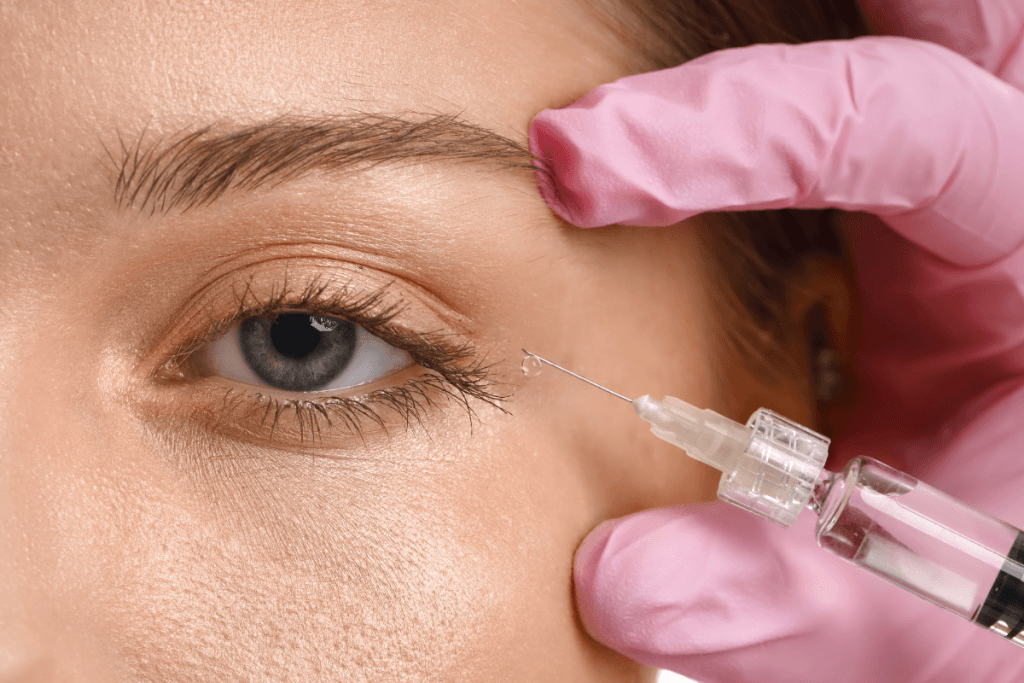
Serious complications associated with intravitreal injection can include endophthalmitis (severe intraocular infection), retinal detachment, intraocular inflammation, and transient rises in intraocular pressure. These are not abstract label items. They directly affect how your team prepares the eye, maintains sterility, performs timeout, documents laterality, and triages post-injection calls. Retina practices also keep class-wide vascular safety considerations in view, even when the immediate operational risk is local to the eye.
More common adverse reactions may include conjunctival hemorrhage, cataract, corneal surface findings, irritation, or other short-term ocular symptoms after treatment. The workflow challenge is that common does not mean trivial. Staff need a consistent script that explains what may be expected, what should be monitored, and which symptoms should trigger same-day review instead of routine reassurance.
Why it matters: Sudden pain, worsening vision, or marked redness after intravitreal injection should not be treated as routine follow-up noise.
For patient counseling, the goal is clarity, not alarm. Clinics typically explain red-flag symptoms such as worsening pain, increasing floaters, light sensitivity, worsening redness, discharge, or a noticeable drop in vision. For staff, the main avoid list is equally practical: avoid using compromised stock, avoid relying on memory for storage exceptions, and avoid normalizing symptoms that may represent infection or inflammation. If you are standardizing office protocols, the Injection Safety hub can help teams compare broader control steps used across office-administered products.
Storage, Handling, and Product Integrity
For clinic operations, storage discipline matters as much as schedule design. The safest workflow keeps refrigeration rules, excursion handling, and vial release criteria in writing rather than in staff memory.
For teams managing Eylea HD in Retina Practice: Label, Safety, Workflow, the refrigerator question should always route back to the live label, carton instructions, and the clinic temperature log. Do not borrow room-temperature rules from standard aflibercept or another anti-VEGF agent. If product sits outside refrigerated conditions, staff should record when that happened, for how long, what temperatures were involved if known, and who approved the final disposition.
The hazardous-drug question also needs a measured answer. In many clinics, this product is handled as a sterile ophthalmic biologic rather than under oncology-style hazardous-drug workflows. Even so, the current labeling, any supplied safety data materials, and local occupational health policy should remain the deciding references. From a practice standpoint, the daily priorities are usually cold-chain integrity, tamper checks, aseptic preparation, sharps disposal, and exposure reporting.
Product integrity checks should be formal. Receiving staff should look at carton condition, expiration, labeling, and any evidence of damage before stock is released. If anything is unclear, quarantine first and resolve later. That approach is safer than trying to fix uncertainty on injection day when schedule pressure is highest.
Clinic Workflow Snapshot
- Verify indication and order before scheduling.
- Confirm carton, vial, and expiration on receipt.
- Log refrigerated storage and any excursions.
- Quarantine damaged or uncertain stock immediately.
- Match product, eye, consent, and lot at timeout.
- Record administration details and follow-up instructions.
- Route red-flag symptoms to urgent review criteria.
Quick tip: Keep room-temperature excursion decisions in a written SOP, not in verbal handoff.
Workflow From Receipt to Chart Closure
The cleanest workflow starts before the patient is in the chair. It begins with supplier review, receiving controls, inventory segregation, and clear documentation of where stock came from and how it was stored.
When a shipment arrives, the receiving step should check product name, presentation, quantity, expiration, and package integrity before inventory is released. If there is a temperature excursion, damage concern, or mismatch between order and delivered stock, the product should be held from use until the issue is resolved under clinic policy. That discipline matters more when a practice carries several ophthalmic biologics with similar names, packaging habits, or refrigerator locations.
Products are sourced through vetted distributors and verified supply routes.
On injection day, staff should reconcile the order, indication, laterality, informed consent, lot number, and expiration before preparation. After administration, the chart should record the product used, lot, date, eye treated, and any immediate reaction. If your revenue cycle team handles buy-and-bill, keep coding references current and validate payer edits regularly rather than relying on a static cheat sheet. Billing and coding are workflow functions, but they still depend on correct clinical documentation at the point of care.
A practical chart review after clinic can catch drift early. Look for missing lot fields, inconsistent indication wording, incomplete post-injection instructions, or storage exceptions that were never closed. Small audit loops often do more to reduce operational risk than a one-time training session.
Comparison Points That Matter Operationally
The most useful comparison is not marketing language. It is which workflow elements actually change when your practice carries more than one anti-VEGF option.
Standard aflibercept and higher-dose aflibercept may share a molecule family, but clinics should still treat labeling, scheduling assumptions, storage references, and templates as product specific. The same caution applies when faricimab or other agents are also on formulary. Operationally, the safer comparison points are indication overlap, documentation burden, follow-up timing assumptions, and inventory separation, not broad efficacy shorthand.
If your formulary also includes Eylea English Alternative or Vabysmo Injection, keep separate refrigerator labels, order sets, and note templates for each product. That reduces look-alike or workflow-drift errors, especially when technicians, photographers, and scribes rotate across different injection days.
Ultimately, Eylea HD in Retina Practice: Label, Safety, Workflow is less about a single injection slot and more about a controlled clinic process. When the label is the reference point, it becomes easier to align scheduling, handling, charting, and escalation decisions across the full visit cycle.
Authoritative Sources
For current labeling and policy detail, these references are the safest starting points.
- FDA prescribing information for EYLEA HD
- Official EYLEA prescribing information from Regeneron
- AAO discussion of anti-VEGF refrigerator management
In day-to-day operations, the safest approach is to make the label the source of truth, then build your refrigerator controls, injection-day timeout, charting, and escalation steps around it. Written workflows are usually more reliable than remembered habits, especially in busy injection clinics.
This content is for informational purposes only and is not a substitute for professional medical advice.