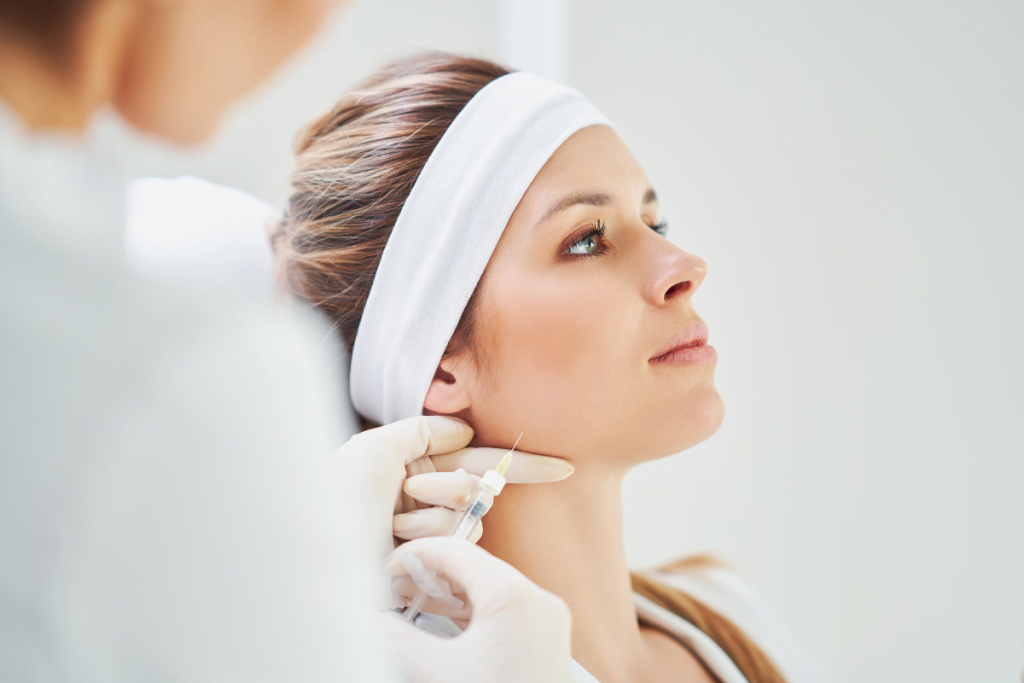
What are Aliaxin fillers? They are a branded family of hyaluronic acid dermal fillers used by trained aesthetic clinicians for volume support, contour refinement, lip work, and soft-tissue correction. The important point for clinics is that Aliaxin is not one universal syringe. Each product in the range should be matched to anatomy, tissue quality, intended depth, current labeling, and the clinic’s adverse-event process.
For licensed healthcare professionals, the practical question is less about brand recognition and more about fit. Two HA fillers can share the same broad category yet behave differently in tissue. That difference affects lift, spread, integration, palpability, and how the result should be reviewed after treatment.
Key Takeaways
- Aliaxin is an HA filler family, not a single product.
- Selection depends on tissue behavior, anatomy, depth, and clinical goal.
- Current IFU and local labeling should guide use, not brand shorthand.
- Clinic workflow should cover consent, traceability, photography, and follow-up.
- Safety planning must include routine reactions and urgent vascular events.
Where Aliaxin Fits in HA Filler Practice
Aliaxin fillers sit within the wider class of hyaluronic acid injectables used in aesthetic medicine. HA is a naturally occurring molecule that binds water. In filler form, it is manufactured as a gel matrix that can add support, occupy space, and improve contour when placed appropriately by trained clinicians.
The manufacturing approach influences rheology, which means how the gel deforms, flows, and resists movement. That matters because a cheek support plan, a vermilion border refinement, and a superficial line correction do not need the same balance of firmness and integration. Clinics reviewing the broader category can use Types Of Dermal Fillers as a category refresher before narrowing to a product family.
In practice, Aliaxin is best understood as a collection. Clinics may encounter product names such as EV, GP, LV, SR, and SV. Those labels can help orient discussion, but they should not replace the current manufacturer instructions for use, local regulatory status, or the injector’s anatomical plan. Product positioning can vary by market, and formulations may be updated over time.
Why it matters: Family branding can hide important differences in gel firmness, spread, and tissue integration.
MedWholesaleSupplies serves licensed clinics and healthcare professionals, so product evaluation should stay aligned with professional procurement, documentation, and treatment governance rather than consumer-style product selection.
Common Aliaxin Filler Uses and Treatment Goals
Aliaxin filler uses generally fall into structural support, contour definition, lip enhancement, and soft-tissue refinement. The exact indication depends on the specific product, the patient’s anatomy, and the labeling available in the clinic’s market.
Clinics often assess HA fillers for areas where age-related volume change, facial proportion, or contour loss affects the treatment plan. Midface support may require a different gel behavior than mobile lower-face refinement. Lip work may prioritize flexibility and integration, while structural contouring may require more projection and support. A single brand family can contain options that behave differently across those goals.
For clinics planning facial balance rather than one isolated correction, Facial Aesthetic Planning offers a useful framework. A visible fold, for example, may reflect support loss elsewhere. Treating only the fold can miss the underlying driver and increase the risk of overcorrection.
Product-specific pages can also help teams understand how a range is organized, provided those pages are used alongside current labeling. Examples include Aliaxin EV Essential Volume, Aliaxin GP Global Performance, and Aliaxin LV Lips Volume. These should support formulary orientation, not replace clinical judgment.
How Clinics Select the Right Product
Product selection starts with the treatment objective. The injector should define whether the case is mainly about projection, contour, line softening, lip shape, asymmetry management, or superficial support. That objective then has to be checked against tissue quality and treatment depth.
Thin skin, scarred tissue, highly mobile areas, and previous filler placement all change the risk-benefit profile. A firmer gel may suit one contouring task but be less forgiving in thin or expressive tissue. A softer gel may integrate smoothly but provide less support where projection is the main priority.
Clinics should also separate named treatment areas from injection planning. Search interest around Aliaxin injection points can make fixed maps look appealing, but safe technique cannot be reduced to memorized dots. Vascular anatomy, depth, prior procedures, and movement patterns all matter. The same product may behave well in one zone and look visible or excessive in another.
| Decision Factor | Why It Matters | What To Verify |
|---|---|---|
| Treatment goal | Lift, spread, and contour needs differ by objective. | Correction, definition, support, or refinement. |
| Tissue quality | Thin or mobile tissue can show product more easily. | Skin thickness, laxity, scarring, and prior filler. |
| Placement plane | Depth affects outcome, visibility, and procedural risk. | IFU, anatomy, training, and local labeling. |
| Patient history | Prior filler or reactions may alter planning. | Previous products, timing, nodules, and delayed swelling. |
| Traceability | Multi-product ranges require precise records. | Lot, expiry, site map, volume, and consent. |
Duration should be discussed carefully. Clinics often ask how long Aliaxin fillers last, but there is no single range-wide answer. Longevity may vary with the specific product, facial movement, treatment depth, metabolic factors, and whether the plan is corrective or maintenance-focused. Set expectations around variability and confirm product-specific information at the point of use.
If the clinic is comparing the range with other HA products, the comparison should focus on treatment role rather than brand preference. The Dermal Fillers Collection can support formulary review, while the editorial Dermal Fillers Articles hub can help teams revisit broader category concepts.
Consultation, Documentation, and Treatment Workflow
A safe Aliaxin treatment workflow begins before any syringe is opened. The consultation should confirm indication, identify risk, define the treatment goal, and establish how the result will be evaluated at review.
Assessment Before Product Selection
Assessment usually includes medical history, current inflammation or infection, active skin disease, bleeding risk, recent dental or facial procedures when relevant, and previous filler history. Permanent filler or unknown prior products require extra caution, especially when the planned plane overlaps the previous treatment.
Photography should be standardized. Baseline images help the team compare swelling, asymmetry, correction, and patient concerns later. They also support internal review if a delayed reaction, product question, or audit issue arises.
Procedure-Day Checks
On procedure day, the team should verify the exact product against the plan, confirm expiry, check packaging integrity, and prepare documentation before treatment starts. The record should capture product name, lot or batch details, treated areas, total volume, anatomical notes, and the immediate response.
Stock governance is part of clinical safety. Clinics should follow current product information for storage and handling, rotate stock by expiry, and maintain traceability across consultation, treatment room, and record system. MedWholesaleSupplies sources brand-name medical products through vetted distributors and verified supply channels for licensed clinics, but each practice still needs its own receiving, storage, and documentation process.
A practical workflow snapshot can keep teams aligned:
- Confirm candidacy and treatment objective.
- Review anatomy and higher-risk zones.
- Check labeling, expiry, and product identity.
- Document consent, photos, and baseline findings.
- Record lot, volume, plane, and site map.
- Provide post-treatment instructions and red flags.
- Schedule review based on clinic protocol.
Quick tip: Keep emergency steps and escalation contacts visible in every filler room.
Safety, Risks, and Contraindications
Aliaxin filler safety considerations are broadly similar to other HA fillers. Expected short-term effects can include redness, tenderness, swelling, bruising, and temporary asymmetry. Less desired outcomes can include visible product, prolonged edema, lumps, nodules, infection, delayed inflammatory reactions, or dissatisfaction with contour.
Contraindications and cautions should be checked against the current product information in the clinic’s market. In general, treatment is often paused or reconsidered when there is active infection or inflammation at the intended site, unstable skin disease, relevant hypersensitivity history, or previous permanent filler in the same plane. Pregnancy, breastfeeding, autoimmune history, anticoagulant use, and recent procedures may also require closer review under local policy.
The serious complication that requires the most disciplined planning is vascular occlusion, meaning blocked blood flow. Although uncommon, it can progress quickly and may threaten skin viability or vision. Higher-risk areas include regions with important vascular connections, such as the nose, glabella, and tear trough. Severe pain, blanching, livedo (mottled discoloration), visual symptoms, or rapidly worsening discoloration require immediate escalation under the clinic’s emergency protocol.
Because Aliaxin is an HA-based filler platform, clinics commonly consider hyaluronidase readiness as part of complication planning where local rules, scope, and training support its use. The key point is preparation. Teams should not wait to develop an emergency pathway after a concerning symptom appears.
Post-procedure follow-through also matters. Mild bruising and tenderness are common. Escalating pain, patchy color change, unusual blanching, fever, drainage, rapidly increasing firmness, or visual disturbance should not be treated as routine aftercare issues. Documentation and prompt reassessment protect both the patient and the clinic.
How Aliaxin Compares With Other HA Filler Options
Aliaxin comparison is most useful when clinics compare treatment intent, not marketing language. HA fillers are not automatically interchangeable because they share the same broad ingredient category. Gel behavior, intended plane, crosslinking approach, and integration profile can all influence how a product performs.
Some HA fillers are selected mainly for support and contour. Others are used where softer integration or more mobile tissue behavior is desirable. Still others sit closer to the skin-quality or hydration end of the spectrum. If the primary concern is diffuse skin texture, elasticity, or hydration rather than structural change, a filler may not be the most appropriate category to consider.
Within the Aliaxin range, clinics may also compare products by role. For example, Aliaxin SV Superior Volume and Aliaxin SR may be reviewed as part of a broader formulary discussion, depending on current availability and labeling. Product names help organize the conversation, but clinical fit still comes first.
Procurement teams should also look at training familiarity, documentation burden, storage requirements, and how each stocked option fits the clinic’s complication protocol. A streamlined formulary helps only when every product has a clear role and the team understands when not to use it.
Authoritative Sources
- FDA overview of dermal fillers
- NHS cosmetic procedure safety advice
- American Society of Plastic Surgeons dermal filler overview
Used well, Aliaxin fillers are part of the wider HA filler toolkit. Clinics should anchor decisions to anatomy, current labeling, product behavior, documentation, and complication readiness rather than relying on product-family names alone.
This content is for informational purposes only and is not a substitute for professional medical advice.