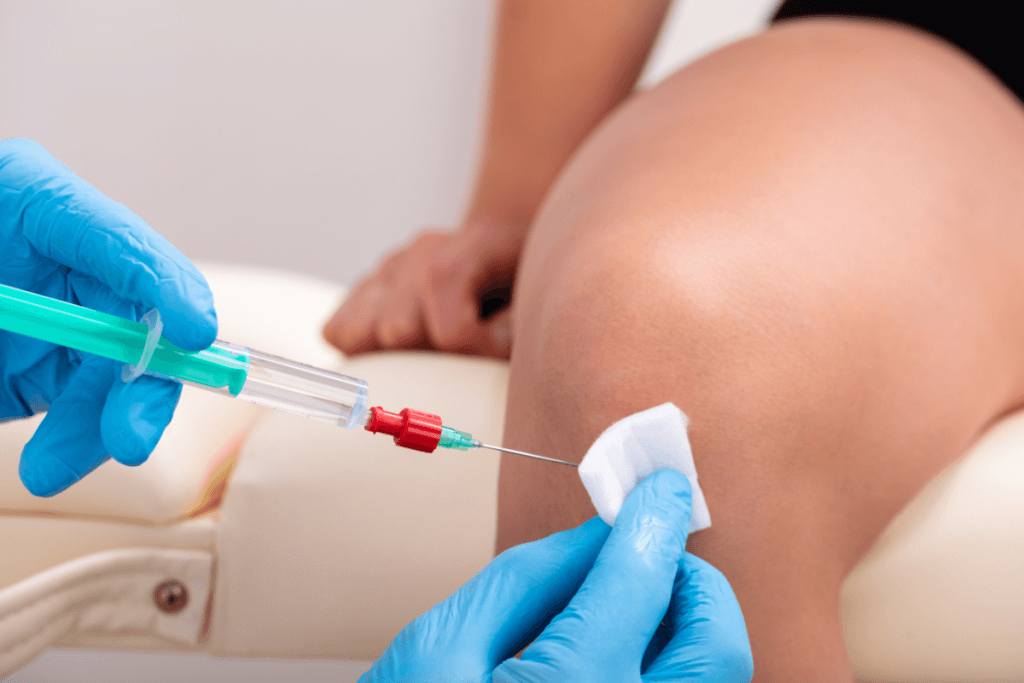
A Monovisc knee injection is a single-injection hyaluronan viscosupplement used in selected adults with knee osteoarthritis (OA) when conservative measures have not provided adequate relief. For clinics, the key question is not whether it is a gel shot in isolation. The practical question is whether the patient, payer documentation, product handling, procedure note, and follow-up plan all fit the same pathway.
This clinic-facing review focuses on that pathway. It explains where the injection fits, what safety checks matter, what patients should be told after the visit, and how teams can reduce documentation gaps before claims or follow-up questions arise.
Key Takeaways
- Single-visit format can reduce scheduling friction, but it concentrates screening and counseling into one encounter.
- Selection should align with current labeling, local protocol, and payer criteria for symptomatic knee OA.
- Expected reactions are often local and short term, while escalating swelling, fever, or severe pain needs prompt review.
- Written aftercare should define activity limits, routine symptoms, red flags, and the callback route.
- Coverage support depends on diagnosis, laterality, prior conservative care, product identifiers, and a complete procedure note.
Where Monovisc Knee Injection Fits in Knee OA Care
This therapy fits as an intra-articular option, meaning it is placed inside the joint, for appropriate knee OA care rather than as a stand-alone cure. Monovisc is a hyaluronan product. Hyaluronan is related to hyaluronic acid, a component of synovial fluid, which helps lubricate and cushion the joint environment. Viscosupplementation aims to supplement that environment for symptom management.
U.S. labeling should guide whether a patient is an appropriate candidate. In general terms, this class is considered after conservative care has been tried and symptoms remain clinically meaningful. Conservative care may include non-drug measures, activity modification, exercise-based management, bracing, weight-related counseling when relevant, or simple analgesic review under the treating clinician’s plan. Exact sequencing varies by payer, facility, and clinician judgment.
The single-injection format is operationally important. It can limit repeat appointment burden, but it also leaves less room to correct incomplete screening later. Before a Monovisc knee injection, the record should already show the diagnosis, treated side, prior management, relevant injection history, and the reason this pathway is being considered.
Selection Questions to Settle Early
- Diagnosis clarity: knee OA is documented with the treated side.
- Prior care: conservative measures are listed in a payer-readable way.
- Clinical fit: symptoms, exam context, and functional limits are recorded.
- Injection history: prior products, response, and adverse events are reviewed.
- Expectation setting: relief is not framed as immediate, certain, or curative.
Why it matters: A strong first-visit record prevents avoidable callbacks and billing rework.
Safety Checks and Contraindications Before the Visit
Safety review should start with the current product instructions, not broad class assumptions. Monovisc safety considerations include local joint reactions, infection screening, hypersensitivity history, and any finding that suggests the knee needs reassessment before routine injection flow continues.
Clinics should screen for active infection or skin disease at the intended site, because injection through compromised tissue can create unacceptable risk. Teams should also review known hypersensitivity concerns related to the product or its components. If the knee is unusually hot, markedly swollen, or clinically atypical, the next step may be evaluation rather than proceeding by schedule.
Bleeding risk, anticoagulant use, recent procedures, or acute injury history may also affect local protocol. These points do not create a universal rule for every patient. They do need a documented decision pathway, especially when multiple clinicians or locations share the same injection workflow.
Pre-Procedure Review Points
- Site condition: skin is intact and clinically appropriate.
- Joint status: no unexpected acute process is suspected.
- Allergy history: product-related sensitivity is reviewed.
- Medication context: bleeding risk questions follow clinic policy.
- Consent language: expected benefits, limits, and risks are documented.
- Product check: packaging, expiration, and storage conditions are verified.
Procedure-day preparation should also confirm that the injector has the current label, approved facility protocol, and required supplies available. Handling should follow the label from receiving through administration. Do not assume all hyaluronan products share identical storage, inspection, or preparation expectations.
Aftercare, Activity Limits, and Red-Flag Symptoms
After a Monovisc knee injection, discharge instructions should be specific enough for patients and staff to use consistently. Many routine questions involve soreness, mild swelling, stiffness, warmth, or a short-lived flare at the treated joint. These symptoms may occur after viscosupplementation, but the chart should define what the clinic considers expected and what requires escalation.
Activity guidance should match the current product IFU and local policy. Official instructions for this product recommend avoiding strenuous or prolonged weight-bearing activity, such as jogging, tennis, or heavy lifting, for 48 hours after administration. That type of language is clearer than telling patients not to overdo it. It also helps triage teams respond consistently when patients call about exercise, work duties, or travel soon after the procedure.
Response timing should also be framed carefully. Some patients may not notice early improvement, and some may experience temporary local discomfort first. Clinics should avoid promising a fixed onset or duration. Instead, document the baseline, give written precautions, and make the follow-up route easy to understand.
When Follow-Up Should Accelerate
- Swelling trend: rapid or worsening joint enlargement.
- Skin change: increasing redness or spreading warmth.
- Systemic signs: fever, chills, or feeling acutely unwell.
- Function loss: new inability to bear weight.
- Pain severity: symptoms out of proportion to routine soreness.
These features do not prove a serious complication on their own. They do justify prompt clinical review for possible infection, bleeding, crystalline arthritis, or another acute joint event. Clinics should decide in advance who receives these calls, how same-day access works, and how escalation is recorded.
Follow-Up Planning and Response Tracking
Follow-up should measure response and safety, not only close the encounter. A useful plan states who will review post-procedure symptoms, when routine outcome checks occur, and how the team will document pain, function, activity tolerance, and adverse events. This matters because viscosupplementation response can vary widely between patients.
The common question about how long relief lasts needs cautious clinic language. Duration should not be promised as a fixed number. Some patients report meaningful benefit for a period, while others have limited or no response. A better workflow is to capture baseline function, record the patient-reported change, and connect that response to the broader knee OA plan.
Repeat-treatment discussions should be policy-aware. Teams should review the current label, payer interval rules, prior response, adverse event history, and any change in diagnosis or joint status before scheduling a new course. That review should happen before authorization work begins, not after the product is already allocated.
Quick tip: Use the same outcome fields at baseline and follow-up to make response review auditable.
Documentation, Coverage, and Clinic Workflow
A Monovisc knee injection can be denied or delayed when the record does not show the indication and pathway clearly. Common gaps include missing laterality, vague prior conservative care, unclear product preference rules, incomplete authorization steps, or absent procedure details. The diagnosis may be accurate, but the claim still needs a record that supports medical necessity and billing requirements.
Useful pre-visit documentation usually includes the diagnosis, treated knee, symptom burden, prior nonoperative care, imaging or exam context when required, previous injection history, consent, and the rationale for the selected viscosupplement. The encounter note should then capture the product administered, lot and expiration tracking when applicable, injection site, tolerance, discharge instructions, and follow-up responsibility.
Procurement review should identify the distributor, verification path, and product identifiers before the procedure day. MedWholesaleSupplies serves licensed clinics and healthcare professionals in a B2B context. Its sourcing model uses vetted distributors and verified supply channels for brand-name medical products.
Clinic Workflow Checklist
- Verify knee OA diagnosis, treated side, and symptom history.
- Confirm prior conservative care and payer-specific requirements.
- Review contraindications, site condition, and recent joint events.
- Check authorization status, product preference, and interval rules.
- Receive, store, and inspect product under current label instructions.
- Document consent, procedure details, product identifiers, and tolerance.
- Give written aftercare with activity limits and escalation language.
- Assign follow-up responsibility for outcomes and adverse event review.
This checklist should be adapted to local protocol. The goal is not more paperwork. The goal is a record that another clinician, biller, or auditor can follow without guessing.
Comparing Viscosupplementation Options Without Brand Hype
Comparison should focus on fit, not brand preference alone. Viscosupplement products may differ by injection schedule, packaging, payer preference, handling details, and documentation burden. A single-visit product may help when repeat scheduling is difficult. A series-based pathway may fit a different formulary rule or practice pattern.
For a series-based comparison point, teams can review Supartz as an example of how multi-visit planning affects appointment slots and chart carry-forward. For broader category browsing, the Joint Injections category can support internal product review. Product pages should not replace label review, clinical judgment, or payer policy.
The most useful comparison questions are practical. How many visits does the pathway create? Is authorization product specific? Can the clinic maintain consistent discharge instructions across locations? Are storage and inspection steps clear to receiving staff? If these questions are answered before the first injection day, the pathway is easier to run and easier to defend later.
Authoritative Sources
- Monovisc instructions for use
- CMS billing and coverage article on hyaluronan injections
- AAOS guideline for knee osteoarthritis non-arthroplasty care
For clinic teams, the best next step is a standardized pathway. Confirm the current label, screen for safety issues, document the indication, give written aftercare, and assign follow-up ownership. That approach keeps the discussion clinical, operational, and easier to audit.
This content is for informational purposes only and is not a substitute for professional medical advice.